Grant Number: P40OD010938
Research Emphasis/Objectives

The Squirrel Monkey Breeding and Research Resource (SMBRR) is a unique national resource that supports research aimed at better understanding the biology of the squirrel monkey, thereby enhancing its value in biomedical research. The SMBRR provides animals, tissues and biological fluids, cell lines, expert advice, and other research resources to NIH-funded grantees.
Current Research
Squirrel monkeys are used in a variety of NIH-supported research programs, including those focused on infectious disease, aging, immune therapies in the treatment of Alzheimer’s disease and related dementias, neuroscience, vision, drug addiction, and substance use.
Services Provided
Preclinical and Regulatory Studies
The Preclinical Studies Program at the Keeling Center for Comparative Medicine and Research serves MD Anderson and other investigators who are developing novel therapeutics and require Good Laboratory Practice (GLP)–compliant toxicology and safety studies to support Investigational New Drug applications to the U.S. Food and Drug Administration (FDA) so that testing in human clinical trials can begin. More than 50 faculty and staff certified in their respective specialty areas from across the Keeling Center support the program. The Keeling Center’s laboratories, animal facilities, and clinical veterinary components are validated for GLPs and have been inspected/audited numerous times by the FDA and outside study sponsors. The laboratories are compliant with the Code of Federal Regulations, 21 CFR Parts 58 and 11. An independent, onsite Quality Assurance Unit reporting directly to the Director of the Keeling Center monitors the program and each individual GLP study.
Anatomic Pathology Services
The Anatomic Pathology Section provides comprehensive diagnostic, collaborative, and research-related pathology services and a tissue/biological fluids banking program. Capabilities include necropsy and histopathology services. In addition, the section conducts FDA-regulated, GLP-compliant nonclinical studies in support of cancer drug development. The staff consists of three board-certified veterinary anatomic pathologists and three certified histotechnologists.
Diagnostic Laboratory Services
The diagnostic laboratory provides traditional laboratory services in the areas of hematology, coagulation, clinical chemistry, microbiology, serology, parasitology, and molecular diagnostics (PCR). The support staff consists of seven medical professionals: five medical technologists (four of whom are certified by the American Society of Clinical Pathologists) and one American Association for Laboratory Animal Science Laboratory Animal Technologist–certified animal resources technologist who supports tissue and biological fluid collection and banking. The clinical pathology laboratory is a validated, GLP-compliant facility; has been inspected by the FDA; and is frequently audited by outside study sponsors. All laboratory services are conducted under the supervision of a veterinary clinical pathologist.
Biological Resources
The Keeling Center is a longtime provider of biological and in vitro products to state and federal government institutions and contract research organizations, as well as diagnostic and academic facilities. The Center specializes in animal whole blood, plasma, serum, tissues, and other products that may be required for clinical testing and research diagnostics. It works to tailor the collection of biological matrices to meet its customers’ needs, including anticoagulant selection, donor population identification, matrix collection protocols, and diagnostic testing. In addition to blood products, other biological materials and tissues may be obtained as requested.
Clinical Laboratory Services
The Keeling Center offers a complete range of clinical pathology laboratory services to support research animal models, discovery research, preclinical studies, and/or clinical trials. Its clinical pathology laboratory is staffed by both registered clinical laboratory scientists and laboratory animal specialists with extensive knowledge of animal and human clinical laboratory services. The Center’s state-of-the-art laboratory can support all stages of human and veterinary drug development, and it conducts testing in accordance with the current standards for GLPs.
Infectious Disease Services
The Keeling Center utilizes a wide variety of animal models in its drug discovery and preclinical research programs. Its Infectious Disease Testing Services are built upon a foundation of contamination prevention measures, effective pathogen detection, and scientific and technical proficiency. The cornerstone of this service is a diagnostic laboratory that delivers sensitive and specific testing to screen animal facilities, research biologics, and a range of research animal models, including mice, rats, and nonhuman primate species.
For more information, please review ORIP’s Nonhuman Primate Resources fact sheet.
Contact Information
William D. Hopkins, Ph.D.
Professor
Keeling Center for Comparative Medicine and Research at The University of Texas MD Anderson Cancer Center
650 Cool Water Drive
Bastrop, TX 78602
Phone: 512-332-7543
Email: [email protected]
Principal Investigator
William D. Hopkins, Ph.D.
Professor
Keeling Center for Comparative Medicine and Research at The University of Texas MD Anderson Cancer Center
650 Cool Water Drive
Bastrop, TX 78602
Phone: 512-332-7543
Email: [email protected]
Center Director
William D. Hopkins, Ph.D.
Professor
Keeling Center for Comparative Medicine and Research at The University of Texas MD Anderson Cancer Center
650 Cool Water Drive
Bastrop, TX 78602
Phone: 512-332-7543
Email: [email protected]
Additional Contact(s)
Elizabeth Lindemann
Keeling Center for Comparative Medicine and Research at The University of Texas MD Anderson Cancer Center
650 Cool Water Drive
Bastrop, TX 78602
Phone: 512-332-5209
Email: [email protected]




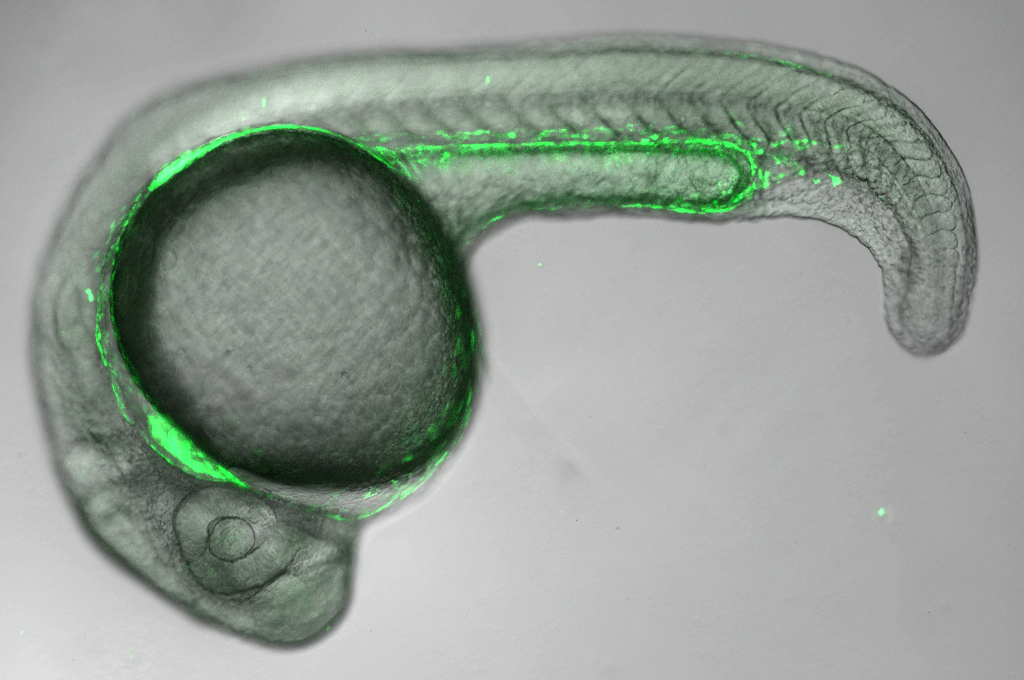
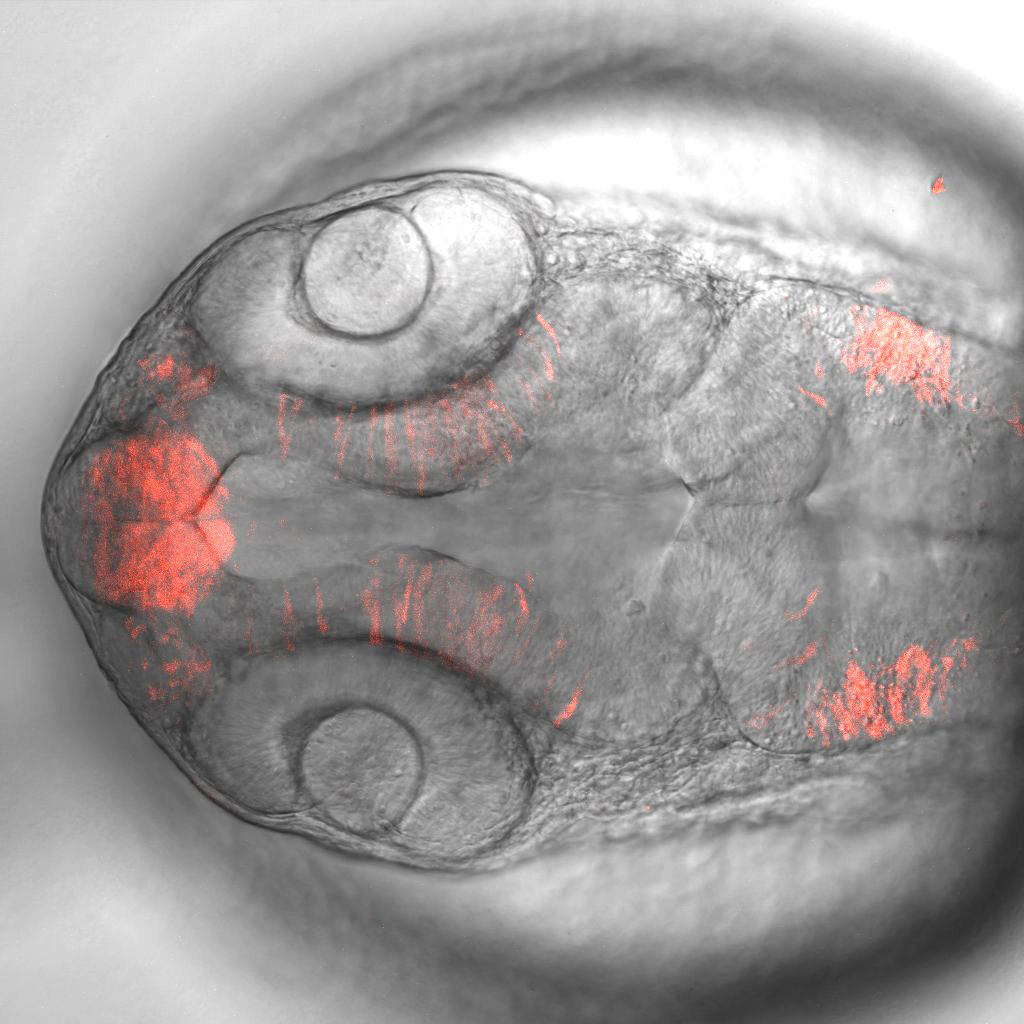 Services Provided
Services Provided

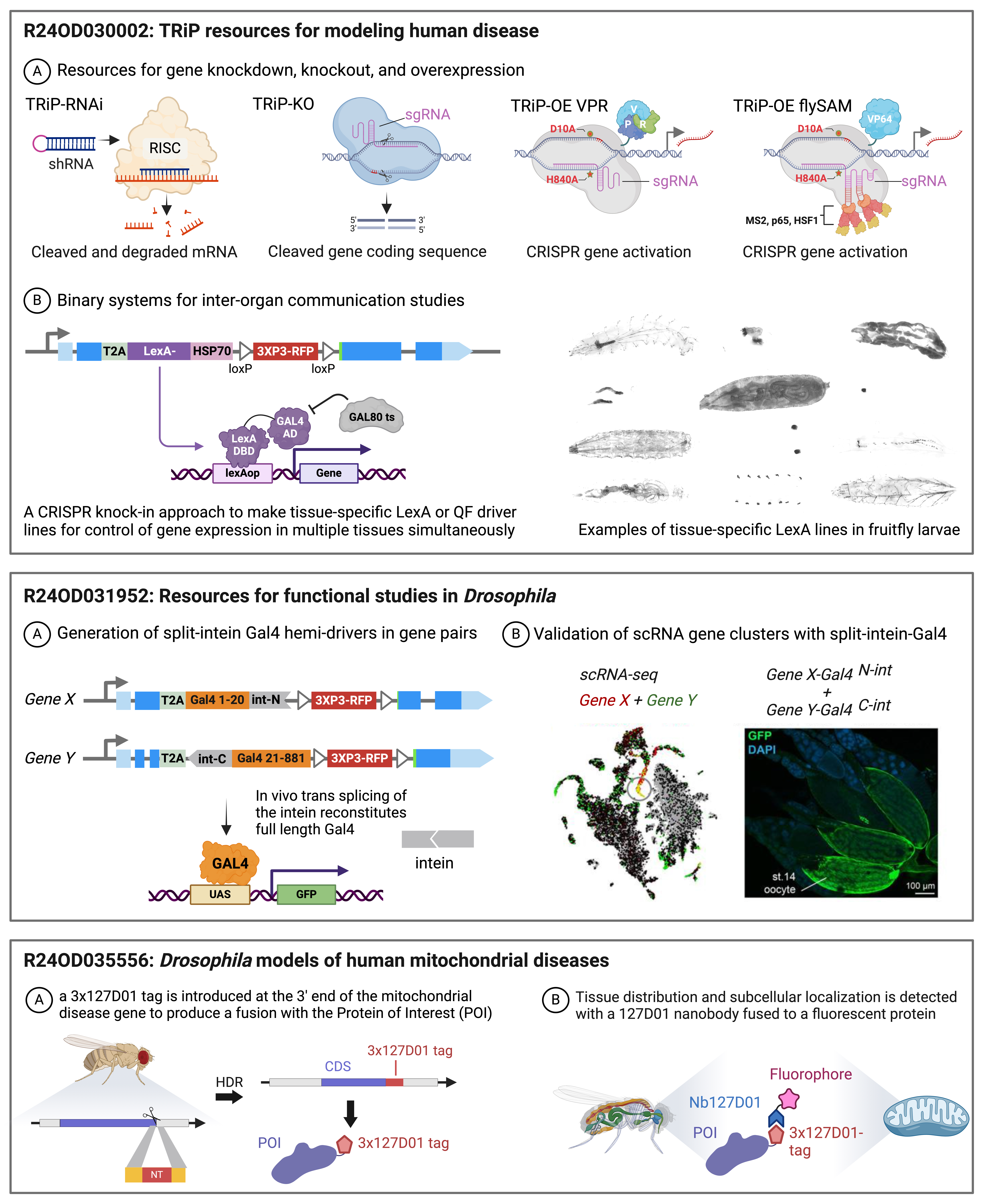 To complement the shRNA and sgRNA collections, new in vivo resources were generated to support three aims. First, the resources enabled combinatorial studies using multiple binary expression systems to study signaling between different tissues and cell types. Focusing on genes with well-characterized GAL4 expression patterns, a set of more than 40 tissue-specific LexA-GAD and QF2 insertions by CRISPR knock-in were generated. Second, the resource enabled the validation of scRNA gene clusters using split-Gal4 lines. Using an algorithm that determines the smallest unique set of marker genes that define a cluster, several hundred highly specific Gal80-repressible split-Gal4 knock-in lines were generated. Last, the resource supported the detection of fly proteins through the insertion of a NanoTag epitope at the C-terminus of more than 300 high-confidence Drosophila orthologs of human mitochondrial disease genes that can then be recognized by an existing high-affinity nanobody against the NanoTag. Together, these new in vivo resources will dramatically expand the scope of reagents available to researchers who are using the fly model to understand the etiology of human diseases.
To complement the shRNA and sgRNA collections, new in vivo resources were generated to support three aims. First, the resources enabled combinatorial studies using multiple binary expression systems to study signaling between different tissues and cell types. Focusing on genes with well-characterized GAL4 expression patterns, a set of more than 40 tissue-specific LexA-GAD and QF2 insertions by CRISPR knock-in were generated. Second, the resource enabled the validation of scRNA gene clusters using split-Gal4 lines. Using an algorithm that determines the smallest unique set of marker genes that define a cluster, several hundred highly specific Gal80-repressible split-Gal4 knock-in lines were generated. Last, the resource supported the detection of fly proteins through the insertion of a NanoTag epitope at the C-terminus of more than 300 high-confidence Drosophila orthologs of human mitochondrial disease genes that can then be recognized by an existing high-affinity nanobody against the NanoTag. Together, these new in vivo resources will dramatically expand the scope of reagents available to researchers who are using the fly model to understand the etiology of human diseases.