S15 Equipment Program: Frequently Asked Questions
![]()
These FAQs provide an overview of the program and its requirements. We always encourage applicants to consult the text of the notice of funding opportunity when preparing an application and reach out to the program contacts with any questions.
Equipment
In general, scientific instruments directly generate scientific data and/or research-essential materials. Examples include microscopes, 3D printers, MRIs, NMRs, mass spectrometers, metabolic cages, and imagers. By contrast, scientific equipment supports and enhances research-related auxiliary activities. This includes glass washers, fume hoods, biosafety cabinets, isolators, and specialized autoclaves/sterilizers. Note, this S15 Equipment Program does not support accessories that are essential for the operations of scientific instruments, such as NMR probes, mass analyzers, or MRI coils. Also not supported are equipment for experimental sample preparation, however, freeze control systems for long-term cryostorage are allowed.
Applicants are strongly encouraged to contact the Scientific/Research Contact(s) of the notice of funding opportunity to determine equipment eligibility prior to the submission of their application.
Applicants are strongly encouraged to contact the Scientific/Research Contact(s) of the notice of funding opportunity to determine equipment eligibility prior to the submission of their application.
No, all requested equipment must support operations of one facility in one physical location.
Telemetry equipment to monitor and improve the management of animal welfare is supported by this program. However, systems that would be used for biobehavioral or physiological assessments as a part of experimental protocols are not allowed.
Yes, for example, various systems that support animal husbandry or monitor environmental conditions in vivaria are supported by this program. This includes systems designed for monitoring conditions in animal cages or systems for environmental controls.
“Free-standing” computers are not allowed. A computer that is integrated into the operation of the equipment is supported. If a term limited (one-year) software license is a part of the request, the application must demonstrate financial support of the software license in outward years.
Program Director/Principal Investigator (PD/PI) and Applicant Institution
An institution that received an equipment award in the immediate prior fiscal year is not eligible to apply for this NOFO unless the previous award has been fully closed out by the date of submission (Sept. 25). In other words, only one active equipment award is permitted at a time.
No, current or past NIH funding is not required to be the PD/PI. Any individual with the requisite knowledge, skills, and resources, necessary to operate and oversee the requested equipment, can serve as the PD/PI.
No, a record of NIH funding is not needed to submit an application and to be considered for an award. However, it is expected that the long term management of the requested equipment and proposed research-related operations will be supported by federal, private, or institutional funding for biomedical research.
The Research Plan should describe plans to support the maintenance and usage of the equipment over the long term (5+ years). Also, a letter from a high-ranking institutional official must be included in the attachments of the application, indicating institutional financial commitment for the operations of the shared facility and research activities conducted therein.
Application Requirements
Basically, a shared facility benefits a relatively larger number of users and is open to the entire institutional biomedical research community, should they need the services that the facility provides and be eligible to receive them (as defined by the institution). This Program does not support equipment that services single investigators.
Yes, please see Section IV in the notice of funding opportunity. In brief, any application must include 1) itemized valid manufacturer’s quote(s) and detailed budget justification, 2) a letter from a high-ranking institutional official, indicating institutional support for the operations of the shared-use facility and research activities conducted therein, 3) a floor plan, indicating the location of the requested equipment, 4) a letter from the Director of Planning, Design, and Construction (or equivalent), and 5) a timeline (of one year duration). An optional table of up to 12 active grants held by end-users of the facility may also be included to demonstrate the need for the added support capacity.
NIH research funding is not a requirement; though if available, it should be demonstrated as a part of the description of the areas of research supported by the facility.
This letter should demonstrate the institution's long-term commitment towards the facility to ensure that the equipment placed there will be used for several years. Specific institutional investments such as personnel costs or other costs associated with running of the facility should be demonstrated.
Cost-sharing is not required or expected. If provided, cost-sharing does not make an application more meritorious. The application budget should be commensurate with the research needs of the equipment, up to the allowable budgetary limit.
Since one page of specific aims is a standard part of the SF424 (R&R) application guide for your S15 application, you should take advantage of it to summarize your project for the reviewers. Bringing the requested equipment into your shared laboratory space or animal research facility should represent an innovative project for which you have goals to enhance the services available to your facility’s users. You may elect to have only one specific aim, stating what equipment is requested and what innovative functionality it will bring. Additional text on this page can be used for a short project overview.
Program Director/Principal Investigator (PD/PI) and Key Personnel
There is no need or expectation to list users of the facility as Key Personnel. In many cases, the PD/PI will be the only Key Personnel listed. In some specific situations, depending on the type of the facility and the equipment requested, an expert in the technology and the facility manager may both be included as Key Personnel. Only one of them can serve as the PD/PI as no Multi-PI applications are allowed under this funding opportunity. In summary, there is not a set number of Key Personnel needed to ensure a meritorious application.
There is no prescribed number of users that would define a facility as shared used. The number of users will depend on many factors, such type of research support offered by the facility through available technologies and expertise in place. Examples include a core facility and an animal facility. Such facilities are typically accessible to the institutional researcher community.
There is not a requisite percentage of biomedical researchers to be eligible, but the requested equipment should provide broad benefits that support and enhance biomedical research activities at the applicant institution.
Application Budget
No, requests for funds to support the alteration and renovation (A&R) of the space the equipment will occupy is not allowed under the notice of funding opportunity.
Funding for the purchase of warranties is not allowed in the application budget. It is expected that the manufacturer of the equipment will provide a 1-year free warranty. However, if a one-year free warranty is unavailable for the requested equipment and the institution declines to purchase it, that piece of equipment is not allowed under the notice of funding opportunity.
The PD/PI must reside at the organization/institution where the requested equipment will be located. Note, funding for equipment is provided to the applicant institution and not the PD/PI who will oversee the use and maintenance of the equipment.
If you place an order before an award is issued, you will not be able to use the award money to purchase the pre-ordered equipment. Moreover, the award (if issued) can only be used to acquire the equipment requested in the application.
S10 Instrumentation Programs: Frequently Asked Questions
![]()
Overview | I. Preparing the Application | II. Submission and Review of the Application | III. Post-Review and Post-Award | IV. Basic Instrumentation Grant (BIG) Program | V. S10 Contacts
Overview
Yes, currently there are three active FOAs:
- Basic Instrumentation Grant (BIG) Program: PAR-22-081
- Shared Instrumentation Grant (SIG) Program: PAR-22-080
- High-End Instrumentation (HEI) Grant Program: PAR-22-079
The S10 mechanism does not directly fund research activities/clinical trials; hence, an S10 grant is not subject to the NIH reporting requirements for clinical trials.
A request for an S10 instrument must be justified by the needs of active NIH-funded research projects. These projects may fund human subject research, including clinical trials. Similarly, it is permitted for an S10-funded instrument to be used for the conduct of NIH-funded human subject research, including clinical trials.
I. Preparing the Application
Instruments and Their Administration
Because of the evolving nature of the S10 program, there is no list of all eligible instruments. Detailed information on types of supported instrumentation and on requests that will not be considered for funding are provided in Section I (Funding Opportunity Description) of the FOAs. We encourage applicants to contact the Program Officer with questions on instrumentation eligibility (see contact info at the bottom of this page)..
Your institution may lease an instrument before the award is received, but only without obligation to buy. If the leasing agreement was executed more than one year prior to submission of the S10 application, the applicant should provide additional information demonstrating that the instrument remains state-of-the-art and the need for requested Federal funds remains. If the application is funded, the award amount will be adjusted based on the remaining balance required to purchase the instrument, as specified in the lease buyout table (which should be included in the lease agreement). The award will not exceed the fair market value of the instrument. An application will be deemed ineligible and eliminated from funding considerations if a purchase order or agreement is executed or a down payment or other formal commitment to purchase the instrument is made prior to award.
Only software that is necessary to control and monitor the operation of the instrument, generate high-quality output data and enable their visualization is allowable under the S10 mechanism. Examples of allowable software include modules to enable special imaging modalities of an optical microscope or pulse sequences for MRI/MRS. In general, software enabling a specific functional configuration of an instrument must be justified by the needs of research projects.
Examples of software items not supported by the S10 program include: (i) separate software items for post-processing of the data; (ii) software supporting data storage and database management; (iii) time- and user-limited software licenses; (iv) duplicate software. In addition, separate workstations and software to operate them are not allowable.
Whenever practical, the S10 funded instrument should be integrated in a centralized core facility, to encourage optimal sharing among individual investigators, research groups, and departments, and to foster a collaborative multidisciplinary environment. If the plans are to place an instrument in a setting other than a core facility or a shared resource facility, the application must demonstrate how the broad access to the instruments will be arranged and the use on a shared basis assured.
Instruments awarded under the SIG program are awarded exclusively for use in biomedical research and cannot be used for billable clinical services.
Under the HEI program, a Special Use Instrument (SUI) application can be considered in rare circumstances. The applicant institution may contribute a portion of the cost of the requested SUI commensurate with the proposed use of the instrument for uses other than biomedical research. The applicants should carefully check the current FOA for details about the SUI requirements. In addition, we strongly encourage the applicants to contact the Program Officer and Grants Management Staff before requesting an SUI as SUIs must meet special administrative rules to be eligible for funding.
In order to improve the cost-effectiveness of the S10 program, the applicants are advised to employ the best economical approaches when planning the purchase and negotiating the price of an instrument, including securing academic discounts, if applicable.
Principal Investigator (PI), Other Users, and Major User Group Requirement
No, the PI of an S10 application does not need to have NIH-funded grant(s) or any other research support. However, the PI should demonstrate in the application that he/she has the relevant instrument expertise and can perform the necessary scientific and administrative oversight responsibilities for the requested instrument.
A Major User is an individual who has a substantial need for the requested instrument, demonstrated by relevant research grant support as a Principal Investigator. Eligible support mechanisms are not limited to NIH grants, but Major Users must be engaged in biomedical research.
A Major User group with at least 3 Principal Investigators on NIH-funded different research awards must be identified (see the answers to the next questions below for additional stipulations).
NIH-funded Major Users should demonstrate a combined need of the instrument at the level of at least 35% of the Accessible Usage Time (AUT). In addition, the projects supported by active NIH research grants should together require 75% or more of the AUT. (See a question in the Other Submission Requirements section about the definition of AUT and related calculations.)
It is not mandatory that a Major User is a PI on an R01 grant. A Major User must be a biomedical researcher with the substantial need for the instrument and commensurate support to fund research using the instrument. An R01 grant is a leading example of an NIH award which offers multi-year support for research activities and may justify funding for the use of an S10 instrument. A PI on an R01 grant may be identified as a Major User. The NIH uses numerous other award mechanisms which offer substantial multi-year funding for the conduct of research; examples include some of the D, K, P, R, and U mechanisms.
The PI of a center grant can be listed as a Major User. However, only one PI of a research-related center grant (such as P01) can be counted as a Major User for the purposes of meeting the eligibility criteria.
Grants supporting infrastructure (e.g., C06), scientific meetings (e.g., R13, R25), training and other non-research activities (e.g., T32, F32) are not eligible to be counted towards the Major User Group requirement. Similarly, SBIR/STTR, Other Transaction (OT) and contract awards are not eligible to fulfil this requirement. PIs who have only NIH awards listed in this paragraph are not eligible to be Major Users. Similarly, these awards cannot be counted towards demonstrating the need at the 75% level of AUT. (For example, a PI on an T32 award is not eligible to be a Major User; however, a PI on an R01 and T32 awards is eligible to be a Major User.)
The need for a specific instrument is demonstrated by the cumulative level of the Accessible User Time (AUT) requested by NIH-funded Principal Investigators. NIH-funded Major Users should demonstrate a combined need of the instrument at the level of at least 35% of the AUT. In addition, the projects supported by active NIH research awards (i.e., grants or cooperative agreements) should together require 75% or more of the AUT.
Other Submission Requirements
Yes:
- A valid itemized quote for the requested instrument must be attached.
- Applications without a quote will be deemed incomplete and returned to the applicant without review (or considered for funding).
- Other budget pages are not required.
- An Advisory Committee must be named.
The membership of the Committee should be broad and include at least one senior institutional official to represent the financial commitments of the institution. Major and Minor Users may be members, but none can chair the committee. The PI cannot be a voting member of the Committee.
Other Submission Guidelines
AUT should be reported in hours per year.
Note that the S10 Program Announcements defines the AUT as “the number of annual hours the instrument can be used for any research purpose”.
NIH recognizes that various instruments require different regular maintenance and calibration schedules. Also, different technologies dictate different experimental timelines or assistance needs of a qualified operator. Hence, AUT hours may be limited by the times an instrument operator is available (if an operator is required), site or building access schedules, estimated or scheduled maintenance, start-up and standardization, and any other factors that take time away from use of the instrument for research.
The AUT for the proposed instrument based on the individual situation at the applicant institution should be described and justified. AUT for the same instrument may differ among different institutions.
For each listed research project specify its anticipated usage in annual hours. (Do not list the same project such as P01 grant multiple times.) Also, include a sum of the anticipated usage in annual hours for all Minor/Other Users – if grouped together. Summarize the usage data in a table. Following the table, state (i) a total estimated usage time by Users’ projects in annual hours, (ii) the percentage of the estimated usage time devoted to Major Users’ NIH-funded projects, and (iii) the percentage of the estimated usage time devoted to NIH-funded projects.
Yes. For-profit organizations are eligible to apply for research awards such as R01 or R21; hence, for example, a PI who is employed by a for-profit organization and is a recipient of an R01 can be a Major User of an S10 instrument. Also, a biomedical researcher employed by a for-profit organization who does not have NIH funding may have access to an instrument funded by an S10 award, depending on specific management and administrative arrangements at the grantee institution. However, NIH-funded researchers have the priority to use an S10-funded instrument.
Justification of Need
No, preliminary data are not required. However, if an instrument can be demoed, including preliminary data is an effective way of showing advantages of the novel technology compared to what’s currently available to the PI and the users.
There is no prescribed number of projects that should be listed in the Research Project Section. The number of users and their projects will vary, depending on the type of the instrument and specific research needs. Important is to clearly demonstrate how each individual research project will benefit from the requested instrument.
Appendix is not allowed. Moreover, links to videos in the body of the application are not allowed either.
Institution Eligibility
No, for-profit, Federal and foreign institutions are not eligible to apply.
Institutional Letter of Support: Table of Previous S10 Awards
S10 grants awarded to the submitting institution (i.e., all departments or divisions with the same DUNS number) must be included in the table listing the previous awards.
As indicated in the FOA, any S10 instruments awarded within the previous five years must be included in the table. For example, for the receipt date in May 2020, the table should list all S10 awards received in FYs 2015 – 2019.
The Table should have 8 columns with the headings as outlined below. For items 6 and 8, please use one of the provided categories:
- Grant Number (e.g., S10OD023456)
- Fiscal Year of Award
- Name of the PI
- Generic Name of Instrument (e.g., Mass Spectrometer, NMR, Confocal Microscope)
- Instrument Status (please enter one of the following options): Active (instrument in use), Pending (order placed but instrument not delivered, instrument received but not installed or not calibrated for general use), Upgraded (or replaced), Not Available (sold, decommissioned, transferred)
- Actual Usage Time (Actual Accessible User Time); Report actual total time in hours per year the instrument was used for research. If the instrument has been installed less than a year ago, the hours can be extrapolated for an estimate of hours per full year.
- Maintenance Agreement (please enter one of the following options): Active (Warranty in place), In-House (or Self-Insured), None (Fee for Service, Pending), Not Available (no longer supported by manufacturer)
- Number of publications that reference this S10 award. Publications that cite the S10 award or publications that have been linked with the S10 award in My NCBI should be included.
| Grant Number | Fiscal Year of Award | PI/PD’s Name | Generic Name of the Instrument | Instrument Status | Actual Usage Time | Maintenance Agreement | Number of Publications Referencing this S10 Instrument |
|---|---|---|---|---|---|---|---|
| S10 OD023456 | 2011 | Jones | Mass Spectrometer | Active | 4000 | Active | 32 |
| S10 OD023788 | 2010 | Lee | NMR | Active | 8000 | Fee for Service | 65 |
| S10 OD023654 | 2014 | Smith | Confocal Microscope | Pending | 2000 | In-House | 96 |
| S10 OD023653 | 2010 | Clark | Robotic Assay System | Upgraded* | N/A | N/A | 74 |
* This instrument was traded-in for an updated robotic assay system purchased with institutional funds.
Supplementary text is only required if the awarded instrument is currently non-functional. Occasionally, other explanatory text may be added if the applicant deems it appropriate (i.e., if there is no maintenance contract since expert self-maintenance is available or if the number of citations is unusually low for the instrument, etc.). Please keep any explanatory text to a maximal length of a sentence or two.
Note that the NIH requires an acknowledgment of an NIH award in each publication, press release, and other documents which report results generated with NIH funding (see the Notice of Award). If there was an oversight in citing the S10 award, the authors should acknowledge NIH support by liking the S10 grant award with these publications in My NCBI.
Institutional Letter of Support to Back Up the Financial Plan
Matching instrument purchase funds are not required by the FOA. However, the institution may decide to provide additional funds towards the purchase of the instrument as part of its institutional support. The institutional commitment letter of support to back-up the financial plan for the operation of the awarded instrument for its expected lifetime is described in the FOA and may include, but is not limited to, preparation of the instrument site, instrument operation, maintenance, and supplies.
It is expected that “the effective lifetime” of an instrument will depend on the type of instrument. The FOA asks an institution for a commitment to back up the financial plan for support of the instrument for five years or “the effective lifetime”. A Study Section will evaluate whether the commitment period is appropriate.
II. Submission and Review of the Application
Submission
We encourage you to submit your application electronically several days prior to the receipt deadline so that you have sufficient time to check the completeness and correctness of your submission, and correct any errors or omissions (if needed). It is not possible to make such corrections after the receipt date has passed.
The S10 program FOAs set a receipt date and the NIH expects that applications will be submitted on time. However, according to NIH Policy for Late Application submissions NOT-OD-15-039, NIH may consider applications submitted within a two week window after the submission deadline but only under the conditions defined in this Notice. The applicant should check the eligibility requirements in the Notice for late submission before considering sending the application late.
Before Review
The Center for Scientific Review (CSR) will convene Special Emphasis Panels (SEPs) within the following Integrated Review Groups (IRGs): Biological Chemistry and Macromolecular Biophysics (BCMB), Bioengineering Sciences and Technologies (BST), Cell Biology (CB), Genes, Genomes, and Genetics (GGG), Interdisciplinary Molecular Sciences and Training (IMST), and Surgical Sciences, Biomedical Imaging, and Bioengineering (SBIB). Applications are assigned to SEPs based on specific instrumentation types. The applicants can access the assignment, the name and contact information of the Scientific Review Officer (SRO), the date of the meeting, and (30 days before the meeting) the roster on their eRA Commons account. Any questions about the SEP assignment should be directed to the SRO.
Late submission of application material is severely restricted. No missing pages or sections will be accepted as late submission materials. Please see NOT-OD-19-083 for additional information.
III. Post-Review and Post-Award
Post-Review
There is no percentile rank for S10 applications.
Do not make an appointment before the release of your application Summary Statement. The Summary Statement, which provides detailed critiques of your application, will be available on the eRA Commons portal within 4 weeks after the review meeting. You are strongly advised to read your Summary Statement before contacting the Program Officer. The Program Officer will not be able to provide information to you prior to release of your Summary Statement.
If you have specific concerns about the review of your application, you are encouraged to contact your Program Officer as soon as you read your Summary Statement and identify concerns to discuss your options. After such discussion, you may choose to submit a written appeal to the Program Officer, signed by the Authorized Official for your Institution. The NIH policy statement on the Appeal Process is defined in the NIH Director Notice NOT-OD-11-064. According to the ORIP procedures, an appeal letter must be received at least 21 calendar days before the Council of Councils meeting at which your application will be reviewed. Around the time of the Study Section, we’ll send you a letter outlining post review procedures and their deadlines.
Just-in-Time notifications are automatically generated by eRA Commons and do not apply to the S10 program. You do not need to submit any information in response to that automated JIT email message. Applicants under consideration for funding will be contacted by Program Officers for updated information from the e-mail address [email protected]. The JIT update should be prepared on the s10reports.od.nih.gov portal that is set up to facilitate collating all required data. S10 updated information includes a (new) valid quote, justification of any modifications of the instrument requested, responses to the weaknesses cited in the summary statement and the need of the instrument to support Users’ research projects. The eligibility requirement to support NIH-funded research projects must be met at the time an application is submitted and when an application is considered for funding, based on the JIT update. This update should also include information about HIV/AIDS related projects, if any.
The use of the portal is strongly encouraged and recommended as it offers many advantages to you, in addition to clearly organizing the required data. You will be able to update and augment the data in JIT to easily create the Final Research Performance Progress Report (Final RPPR, FRPPR) and four subsequent Annual Usage Reports, as defined in the Notice of the Award.
Typically, an award is made at the level approved by the Study Sections, provided that other budgetary constraints are met.
The applicant may consider resubmitting an application, after addressing the reviewers’ comments. Alternatively, the applicant may submit a new application. In such case the narrative must not refer to the previous review.
Post-Award
A letter signed by the current PI and Authorized Institutional Business Official, including reasons for the change and the proposed effective date of change, and the biosketch of the new PI are needed to initiate the process. These documents should be sent to the Grants Management Specialist and copied to the Program Officer listed on your Notice of Award.
The PI must contact the Program Officer and the Grants Management Specialist assigned to the award to discuss the possibility of relocation of the instrument. The Internal Advisory Committee, as well as both institutions involved, must approve such relocation. Considerations must include the needs of the user group noted in the application and those of the proposed new user group at the accepting institution.
Pre-approval from both the Program Officer and the Office of Grants Management is required for the following changes: relocation of the instrument inside the institution (i.e., assigned to a different core facility or different school within the institution); relocation of the instrument to another institution; change of PD/PI; trade-in, sale or other disposition of the awarded instrument; shut-down of an instrument within less than five year of its installation. Any of these changes also require approval of the Internal Advisory Committee for the instrument.
Since FY2006, any S10 Notice of the Award has included the language: “Each publication, press release or other document that cites results from NIH grant-supported research must include an acknowledgment of NIH grant support […]”. Per NIH-wide policies, any publication that reports data collected using an S10 instrument must be linked to the S10 award using MyNCBI (like it would be done for any research grant, for example an R01) or acknowledge the award in the body of the paper.
An S10 award is issued for a year. Within that period, an instrument should be ordered, delivered, calibrated, and made available to researchers. The Final Research Performance Progress Report (FRPPR) must be submitted at the project end period when the instrument is fully functional and operational. As any instrument is expected to be used for many years subsequently to FRPPR, four Annual Usage Reports (AURs) are required, as defined in the Notice of the Award. FRPPR and AURs should include a report from the Advisory Committee.
IV. Basic Instrumentation Grant (BIG) Program
General
The program targets institutions that receive lesser amounts of NIH funding per year compared with leading biomedical research institutions in the US. This opportunity can, thus, make a significant (BIG) impact on institutions with somewhat limited resources. For these institutions, acquisition of modern-day costly instruments may contribute to: (1) advancement of important biomedical research projects at locations with limited extramural funding or (2) development of new biomedical research projects at these institutions.
Since the BIG program supports acquisitions of basic instruments, funding is limited to $25,000 - $250,000.
Shared instrumentation programs managed by ORIP are open to public and private institutions of higher education as well as nonprofit organizations with or without 501(c)(3) IRS Status. However, the BIG program restricts eligibility to only those institutions that have not received S10 funding greater than $250,000 in any of the past three fiscal years – hence, limited competition.
For example, if the receipt date for BIG applications is in calendar year 2021 (June 1, 2021), you should check whether your institution has received any S10 awards in fiscal years 2018-2020. For BIG applications with a receipt date in calendar year 2022, the eligibility would be determined by whether your institution received S10 awards in fiscal years 2019-2021, and so on. A table on the ORIP website (https://orip.nih.gov/construction-and-instruments/s10-instrumentation-programs/filterable-awards-table) can provide you with such information. However, your institution should confirm eligibility by checking the NIH RePORT website (https://report.nih.gov/award/index.cfm). Note, an application submitted for the BIG program must include a letter from an Authorized Organizational Representative confirming eligibility.
For example:
- If an institution received an S10 award of $150,000 in FY2018 and another S10 award of $180,000 in FY2020, the institution is eligible to apply for the receipt date in 2021 – because for any year in FYs 2018-2020, the institution did not receive an S10 award greater than $250,000.
- If an institution received two S10 awards $200,000 and $125,000 in FY2019, the institution is not eligible to apply for the BIG receipt date in 2021. In this situation, the combined total of S10 awards in FY2019 exceeds the limit of $250,000.
- If an institution received two S10 awards in FY2019 of $100,000 and $125,000, the institution is eligible to apply for the receipt date in 2021 since the combined total of S10 awards in FY2019 is less than $250,000.
An institution – as identified by its DUNS number, can submit only one application to the BIG program per receipt date. However, in addition to one BIG application, an institution can submit (multiple) applications to the SIG and HEI programs, for different types of instruments/technologies – please check the text of the corresponding Funding Opportunity Announcements for a detailed description of the conditions under which multiple applications from an institution are allowed.
The shared instrumentation program provides scientific instruments to advance NIH-funded research and as such the BIG program meets this objective. The BIG program targets institutions that are not major recipients of NIH research funding and thus, the requirement of NIH research funding for BIG instrument users is defined according and somewhat differently than for the SIG and HEI programs. Specifically, users whose project are supported by grants such as R03, R15, or R21 qualify as Major (and Minor) Users for the BIG program. Multi-year funding demonstrated by grants such as R01s, is not required though is allowed and accepted by the BIG program. Note, like the SIG/HEI, the BIG program requires 3 Major Users with NIH research funding. Training or fellowship grants (i.e., T and F mechanisms), other non-research and SBIR/STTR grants, Other Transaction (OT) awards, and contracts cannot be used to fulfill the Major User requirement.
First, as for any S10 shared instrument, the PI should determine the accessible usage time (AUT) which is the number of annual hours the instrument can be practically used for biomedical research. The projects of Major Users supported by any NIH research awards must collectively use the instrument for at least 25% of the AUT. Moreover, the total collective usage for biomedical research projects (irrespective of funding source) for all identified users (Major and Minor) should be 75% of AUT.
The receipt date for ORIP-managed S10 programs - BIG, SIG, and HEI, is the same; typically end of May/beginning of June each FY. Please check the Funding Opportunity Announcement for the receipt date, and the timeline for peer and Council reviews, and funding considerations.
V. S10 Contact
Monika Aggarwal, Ph.D.: [email protected]
Division of Construction and Instruments (DCI)
Office of Research Infrastructure Programs (ORIP)
Division of Program Coordination, Planning, and Strategic Initiatives (DPCPSI)
The Office of the Director, NIH (OD)
6701 Democracy Boulevard
Bethesda, Maryland 20892-4874
Phone: 301-435-0783
C06 Program: Frequently Asked Questions
Eligibility
Domestic public or private nonprofit institutions of higher education and nonprofits other than institutions of higher education are eligible.
Hispanic-serving Institutions, Historically Black Colleges and Universities (HBCUs), Tribally Controlled Colleges and Universities (TCCUs), Alaska Native and Native Hawaiian Serving Institutions, and Asian American Native American Pacific Islander Serving Institutions (AANAPISIs) are encouraged to apply.
Yes, NIH intends to commit at least 25% of the available budget to support meritorious projects from Institutions of Emerging Excellence (IEEs).
Typically, IEEs receive low levels of NIH funding to support basic, clinical, and translational research. Often, IEEs are in geographical areas with deficits in research resources. Likewise, IEEs often serve individuals from disadvantaged backgrounds. IEEs play a special role in advancing biomedical research by leveraging their research capabilities to address problems of special relevance or unmet health needs. For the full description of the characteristics of IEEs, consult PAR-23-045, USC 42 Section 283k, and USC 42 Section 293.
No. All domestic institutions of higher education (state-controlled and private nonprofit) and nonprofits other than institutions of higher education are eligible.
Yes. The construction of animal facilities is allowable under this funding announcement.
The PD/PI should be a PhD-level administrator or senior scientist who has appropriate stature within the institutional administration structure to oversee and bring to completion the various facets of the entire project. The PD/PI should also demonstrate leadership skills to recruit and guide expert Key Personnel with complementary scientific and technical skills to successfully plan, design, and carry out the construction project. The PD/PI does not need to be NIH funded at the time of application submission.
Key Personnel must include an individual with engineering /architectural expertise (Construction Project Manager) who will communicate with investigators / facility users about the research-driven infrastructure needs and who will work with an outside engineering firm designing the facility. In addition, depending on the scope of the project, Key Personnel may include subject matter experts on specialized research infrastructure needs for their research (Facility Manager); for example, a veterinarian may offer useful expertise if an animal facility is proposed, or a drug development and compounding expert may be relevant if a pharmacy facility to support human clinical trials is proposed.
Yes, but you must submit as a new (A0) application. Do NOT include an introduction or discuss the previous submission(s) in the A0 application. Failure to comply may result in withdrawal of the application from review.
Projects
Yes, proposals to modernize and improve existing research facilities (e.g., laboratory space, core, or animal facilities), or to construct new biomedical research space and furnish it with necessary fixed equipment are supported. Construction of unfinished shell space is not allowed.
Any project must be justified by needs of a broad research community, locally at the applicant institution or regionally. When completed, any project should offer long-term benefits by providing a modern research environment, accessible on a shared basis.
Inappropriate requests are for:
- regular maintenance, replacement of aging or failing equipment, and other routine work;
- facilities for billable medical care, office space, or classrooms;
- upgrades of a space serving a single investigator.
Inappropriate requests will be not be considered for funding.
A successful project will create a space serving multiple researchers from the applicant institution or neighboring institutions. Examples include construction of space housing a new core facility or a project consolidating core facilities to support an array of services. Likewise, remodeling of laboratory space serving college- or department-wide to support specialized workflows and environments (e.g., an animal surgical suite, a gnotobiotic barrier facility, space supporting biobehavioral studies linking observations, imaging, and molecular characterization) is another relevant example.
No, any project should be consolidated; for example, located in the same building and composed of contiguous space.
Projects that serve the institutional-wide biomedical research community are allowed. It is expected that every project will strengthen biomedical research programs, by bringing novel capacities to research infrastructure.
Yes, but the proposed project must be clearly marked and distinguishable from the larger non-application part of the overall plan.
Budget
The budget should reflect the scope of the construction project and must be commensurate with institutional needs to upgrade biomedical research infrastructure. Awards will be issued in the range from $3 million to $8 million. Applications requesting projects with a budget of less than $3 million will not be considered.
Cost sharing is not required or expected. If a proposed project is larger than the maximum award of $8 million, an institution may contribute non-federal funds towards the project. Contributing institutional funds does not make any request more meritorious.
Fixed equipment are assets that are attached and integral to the building’s structure and function. Building services equipment or systems, such as heating, ventilation, air-conditioning (HVAC), elevators, electrical plumbing, and water sprinkler systems are examples of fixed equipment. Also, equipment permanently attached to such building services or systems, necessary to enable technical functions, are generally allowable as fixed equipment. Examples of such fixed equipment include fume hoods, aquaculture system (including plumbing, filtration, pumps, and aquarium racks), walk-in cold-rooms, and cage/rack washers. However, plugging movable equipment into an electrical receptacle of a building does not confer on it a fixed equipment status. For example, freezers and refrigerators (typically on wheels) are not fixed equipment. Scientific instruments are not allowable within construction awards.
Yes, the proposed construction project must be fully functional and operational at the completion of construction and occupancy.
Yes, funds may be requested for select space within a larger facility to be renovated.
No, only direct costs for the construction project are allowed. Facilities and Administration (F&A) cost are not allowed.
No, any costs incurred before the issuance of a Notice of Award may not be reimbursed or otherwise charged to the grant.
Applications
The narrative page limit is 13 pages -- 1 page for the Overview section and 12 pages for the Scientific Justification and Facility Development section. Additional line drawings and attachments which are required do not count towards this 13-page limit.
The narrative should explain why biomedical investigators at the applicant institution needs the proposed facility/laboratory space and how the applicant institution will use it to advance their research. Research needs should drive the proposed engineering solution and the application. Hence, the narrative should include a description of the technical design justifying the proposed engineering solutions and how they will meet the research needs.
Research-intensive institutions should link the proposed construction projects to currently received NIH funded research awards and document institutional commitment for supporting research in these areas of science. There is no requirement of having secured NIH research funding for submitting an application. IEEs should detail long-term institutional strategic plans for strengthening biomedical research programs and for support of identified areas of special need. Available expertise and projected growth should also be outlined.
Yes, required attachments include: the Overview section, Scientific Justification and Facility Development section, Biosketches for the PD/PI and Project Manager, Annotated Satellite and/or Map Views of the Site, Line Drawings, Table with list of rooms/components including dimensions, Table of requested fixed equipment, Project Timeline, Budget Justification and Vendor Quotes, an Institutional Support Letter from a high-ranking institutional official, and Verification of Title to the Property - check the FOA for the complete list of required attachments.
No, the line drawings do not count towards the page limit, but do not include additional text on the drawings to circumvent the limit. Line drawings are not “blueprints”; each drawing must fit on an 8.5”x11” sheet of paper.
If a state does not have a Single Point of Contact (SPOC), select NO to question #16, box “Program is not covered by E.O.12372.”
No.
No, you should not use Field #8 for uploading your main project description. Field #8 is to be used for a very short 3-4 sentence description of the public health relevance of your proposal. The Overview (1-page) and Scientific Justification and Facility Development (12-pages) sections, should be the first two “Other Attachments” as described in the FOA under Other Project Information. Please also review Page 6 of the Annotated Forms that can be found at the following link: https://grants.nih.gov/grants/ElectronicReceipt/files/Annotated_Forms_General_FORMS-H.pdf.
Technical Design
The project design should follow federal, state, and local standards and codes for biomedical research facilities so that upon the completion of this project, the occupancy permit can be secured, and the facility can serve its research purpose. The NIH Design Requirements Manual (DRM) is a useful document that provides design guidelines for NIH-funded construction projects.
The use of modern engineering solutions and technologies is encouraged to make the constructed space compatible with the infrastructure needs of the state-of-the art research projects. Especially encouraged are the applications of green technologies offering efficient energy use, the reduction of waste, and the elimination of hazardous substances. Applicants are invited to follow sustainable design principles, leaving a minimal imprint on the environment.
The required line drawings which are part of the application, are not “blueprints” or complete design documents. Nevertheless, the drawings should be of professional-level quality (e.g., to standards used by engineers or architects), to clearly indicate dimensions and functions of the areas affected by the project. The drawings should include details such as locations of equipment, safety clearances, egress routes, or changes/additions to existing mechanical and electrical systems. Do not add additional narrative on the drawing to circumvent the 12-page limit for the Scientific Justification and Facility Development Section.
Only fixed infrastructure equipment is allowed. Examples of such equipment include: fume hoods, surgical lights and procedure tables, ventilated racks, HVAC systems, water purification systems and so forth. Scientific instruments (e.g., mass spectrometers, microscopes, biomedical imagers) are not allowed.
Post-Submission
Applications will be reviewed for scientific merit by a study section whose membership will include PIs’ peers (researchers/administrators) and engineers/architects with expertise in the design of biomedical facilities. Merits of each application will be judged based on the review criteria listed in the FOA:
- Scientific Merit and Organization of the Total Program and Its Component Parts to be Carried Out in the Facility;
- Administrative and Leadership Capabilities of the Applicant’s Officers and Staff;
- Anticipated Effect of the Project on Other Relevant Research Programs and Facilities in the Geographic Area and Nationwide;
- Need for the Project or Additional Space;
- Project Design.
We encourage potential applicants to go over the description of these criteria in the FOA and to be sure to include in the application all relevant information upon which the application will be peer-reviewed.
Following review by a study section, the applications will undergo a second level of review by the Council of Councils (CoC). The CoC is composed of both scientific and public representatives chosen for their expertise, interest, or activity in matters related to health and disease.
Only applications that are favorably recommended by both the study section and the Council of Council may be recommended for funding. Funding decisions are based on the outcome of peer review, relevance to the program priorities, portfolio balance, and budgetary considerations, among other factors as described in the FOA under "Review and Selection Process".
The terms of the Notice of Award will state that only funds for the design of the construction project will be available immediately following the issuance of an award. The non-design funds are restricted until NIH approves the final construction design documents.
For awarded projects, design documents may be submitted to the NIH for technical review immediately following the issuance of the Notice of Award. Generally, there are three design document submissions. It is possible to submit a combination of schematic and design development documents in the first submission (about 60% completions) or 95 to 100% completed construction documents.
Other
Funds for NIH extramural construction requires a specific Congressional appropriation. When funds are available, a funding opportunity announcement will be published in the NIH Guide for Grants and Contracts.
Institutional Research Training Grants
List of States by Institutional Award
University of California, Davis
Major Areas of Interest

Biomedical research, especially infectious disease, cancer, genomics, and animal models of human disease
The mission of the Comparative Medical Science Training Program (CMSTP) is to prepare post-D.V.M. Ph.D. students to become exceptional veterinarian-scientists in comparative medicine at the University of California, Davis (UC Davis). This postdoctoral T32 is highly successful in training veterinarian-scientists for research careers, with more than 90% utilizing their research training.
To ensure the success of our new trainees, we have a comprehensive 4-year program at UC Davis, which supports D.V.M.s in pursuit of their Ph.D. We will prepare trainees for the next stage of their careers through provision of research colloquia, individualized mentoring, networking opportunities, and grant writing workshops. Thus, CMSTP provides a comprehensive, integrated research-mentoring program for D.V.M.s at UC Davis, particularly those from historically marginalized and disadvantaged backgrounds, to remove barriers that exist and deter some from pursuing a research career. We are committed to recruiting and retaining a diverse body of trainees.
We have 36 highly qualified faculty trainers to choose as a mentor or a co-mentor if the trainee has already chosen a lab. The CMSTP takes advantage of outstanding research and teaching opportunities in the biological sciences at UC Davis by bringing together faculty from the School of Veterinary Medicine, the nation’s top veterinary school; School of Medicine; College of Engineering; and College of Biological Sciences. Our faculty trainers participate in established Centers and Institutes that promote collaborations and employ diverse evidence-based approaches to solving scientific problems through state-of-the-art equipment in individual labs, as well as campus shared facilities.
The objectives of the CMSTP are to (1) prepare all of our trainees to become future leaders in academia, government service, public health, and biomedical research; (2) provide greater exposure to career paths outside academia; (3) maintain the average time of 4 years to degree; and (4) attract and train a diverse group of veterinarian-scientists.
Contact Information
University of California, Davis
Center for Comparative Medicine
One Shields Avenue
Davis, CA 95616
https://www.vetmed.ucdavis.edu/research/cmstp
Grant Number: T32OD011147
Program Director
Sara Thomasy, D.V.M., Ph.D., DACVO
Phone: 530-752-1770
[email protected]
Colorado State University
Title
Biomedical Research Training for Veterinarians
Major Areas of Interest

- Prion biology
- Immunology
- Mycobacterial pathology
- Retroviral research
- Vector-borne and infectious disease
- Cancer biology and diagnostics
- Reproductive technology
- Orthopedic research
- Pathobiology
- Translational and comparative medicine
Contact Information
Grant Number: T32OD010437
Program Co-Director
Kelly Santangelo, D.V.M., Ph.D., DACVP
Phone: 970-491-5015
[email protected]
Johns Hopkins University
Major Areas of Interest
Infectious diseases, immunology, cancer biology, neuroscience, experimental pathology, genomics, translational animal models, data science
Based at a leading academic medical center, the Johns Hopkins Training Program provides outstanding biomedical research training opportunities for veterinarians interested in careers as veterinary scientists. The training program is based in the Department of Molecular and Comparative Pathobiology, a basic science department linked to our comparative medicine mission in animal care including clinical lab animal medicine and comparative pathology. Ph.D. and postdoctoral research training can be performed in a wide variety of disciplines across the university and may be combined with complementary training in lab animal medicine or anatomic pathology.
Contact Information
Momina Malik
[email protected]
Grant Number: T32OD011089
Director
Joseph L. Mankowski, D.V.M., Ph.D.
Phone: 410-955-9770
[email protected]
University of Minnesota
Major Areas of Interest
The Comparative Medicine and Pathology Training Program provides up to 3 years of support for veterinarians to participate in state-of-the-art biomedical research training with the goals of providing individuals with the broad-based knowledge, quality communication skills, and advanced research training essential for a career as a veterinarian scientist. The robust training program, which is housed in the College of Veterinary Medicine, is uniquely positioned to perform cooperative, translational research with other colleges at the University of Minnesota, including the Medical School; School of Public Health; College of Biological Sciences; School of Dentistry; College of Food, Agricultural, and Natural Resource Sciences; and College of Education and Human Development. This ensures a steady supply of interdisciplinary opportunities for faculty and students to promote “One Health.” Close connections with these other colleges have enriched the opportunities of current and past trainees, allowing them to work with faculty mentors, thesis committee members, and instructors representing a wide range of disciplines, including emerging and zoonotic infectious disease, pathology, pharmaceutics, genetics and genomics, comparative and translational medicine, public health, oncology, immunology, orthopedics, and epidemiology. The diverse interests of faculty, centers and institutes, and graduate classes allow trainees to tailor their curricular and professional development activities to meet their individual training needs. Past trainees have successful careers as academic, government, and private-sector researchers. A strong alumni connection with former fellows enriches the training experience through near-peer mentoring to trainees.
Contact Information
Office of the Associate Dean for Research
College of Veterinary Medicine
University of Minnesota
225 Veterinary Medical Center
1365 Gortner Avenue
St. Paul, MN 55108
https://sites.google.com/umn.edu/dvmt32/home
Grant Number: T32OD010993
Principal Investigator
Molly McCue, D.V.M., M.S., Ph.D., DACVIM
Phone: 612-624-9320
Fax: 612-624-8753
[email protected]; [email protected]
University of Missouri-Columbia
Major Areas of Interest
Research opportunities are available in more than 40 well-funded research laboratories in a broad range of areas related to comparative medicine research including, but not limited to, studies in genetics of disease; impact of microbiome; disease pathogenesis; genetic engineering; immunobiology; cardiovascular, renal, and neurological function; cancer research; cryobiology and assisted reproduction; and biomedical engineering. Furthermore, the presence of several NIH-funded animal Resource Centers on campus, including the MU Mutant Mouse Resource and Research Center, Rat Resource and Research Center, National Swine Resource and Research Center, and one of 12 NIH-funded regional biocontainment laboratories provide a unique training environment for research, development, and characterization of genetically engineered animals.
The MU Comparative Medicine Training Program (CMP) provides a substantive foundation for a competitive research career through (1) coursework and seminars that give a broad exposure to biomedical sciences, comparative medicine, statistics, and experimental design; (2) an intense research experience to provide competence in state-of-the-art experimental methodology; (3) instruction in fundamental concepts of funding procurement and development of grant-writing skills; (4) instruction in all aspects of lab and project management including development of transferable skills; (5) instruction in scientific rigor and transparency in experimental design and study reproducibility and translatability with an emphasis on animal modelling; (6) training in the ethical conduct of research; and (7) increasingly independent experience in every stage of the scientific research process. This training enables CMP graduates to initiate careers in research, become leaders in the field of comparative medicine, and fill the critical void of veterinarians in biomedical research.
Contact Information
Comparative Medicine Program
University of Missouri
Department of Veterinary Pathobiology
College of Veterinary Medicine
4011 Discovery Drive
Columbia, MO 65201
Grant Number: T32OD011126
Program Directors
Craig L. Franklin, D.V.M., Ph.D., DACLAM; Elizabeth C. Bryda, Ph.D.
Phone: 573-882-6623; 573-882-5504
[email protected]; [email protected]
Cornell University
Major Areas of Interest
The Cornell Graduate Training Program in Comparative Medicine combines the very best that Cornell offers in the form of didactic graduate-level instruction, faculty supervision, and training related activities. Trainees can follow one of two tracks: one is geared to a career in basic research and a second to a career in translational science. Training is structured to ensure the orderly progression of scholars to independence. Research areas available to trainees are broad and include infectious disease, immunology, epidemiology, cancer biology, cell biology and signal transduction, genomics and genetics, developmental biology, molecular medicine, biomedical engineering, and neuroscience.
The program combines independent, faculty-guided research with formal didactic instruction in cell and molecular biology, genomics, and biostatistics; career counseling; and a variety of professional enrichment activities calculated to develop the trainees’ critical capacity, communication, and teamwork skills. Graduate scholars would earn the Ph.D. degree. The average time to degree for veterinarians seeking a Ph.D. at Cornell University is 4.3 years. A Graduate Research Assistantship provided by the College of Veterinary Medicine at Cornell provides stipend to all scholars during the first 9 months of the Ph.D. while trainees are completing three laboratory rotations.
Almost all Program alumni go on to obtain faculty positions or positions in industry or government. The mission of our program is to train veterinary scientists that can meet the national need for trained veterinarians within academia, industry, public health, and government to address old and new problems relating to animal and human health.
Contact Information
Grant Number: T32OD011000
Program Directors
John S. Parker, B.V.M.S., Ph.D.; Robert Weiss, Ph.D.
[email protected]
Wake Forest University
Major Areas of Interest
The Department of Pathology, Section on Comparative Medicine, of Wake Forest University School of Medicine offers four postdoctoral fellowships in comparative medicine. These positions provide a unique opportunity for motivated graduate veterinarians interested in advanced biomedical research training leading to an independent research career. The fellowship typically leads to a Ph.D. degree.
Research opportunities include internationally recognized programs in primate research, aging, women’s health, cardiovascular disease, cancer biology, radiation biology, regenerative medicine, diabetes, reproductive system disorders, and substance abuse.
The 27 training faculty committed to the program represent a variety of basic and clinical disciplines. The broader school of medicine community consists of more than 1,200 faculty comprising 41 academic departments and 26 Centers or Institutes. Major institutional resources include the Clinical and Translational Science Institute, national primate research resources, Wake Forest Baptist Comprehensive Cancer Center, Hypertension and Vascular Research Center, Wake Forest Institute for Regenerative Medicine, and Sticht Center for Healthy Aging and Alzheimer's Disease Prevention.
Institutionally supported training programs in laboratory animal medicine and veterinary anatomic pathology are also available.
Contact Information
Wake Forest University School of Medicine
Pathology/Comparative Medicine
Medical Center Boulevard
Winston-Salem, NC 27157-1040
Grant Number: T32OD010957
Principal Investigator
J. Mark Cline, D.V.M., Ph.D., DACVP
Professor of Pathology/Comparative Medicine and Radiation Oncology
Phone: 336-716-1564
Fax: 336-716-1515
[email protected]
Texas A&M University
Title
T32 Comparative Biomedical Research Training for Veterinarians (CBRT)
Major Areas of Interest

Biomedical research: biomedical genomics and bioinformatics, diagnostics and therapeutics, infection, immunity and epidemiology, physiology and developmental biology
Major research areas include infectious and metabolic diseases, toxicology, nutrition, developmental biology, discovery of therapeutic molecules, cancer biology, and neuroscience.
The T32 Comparative Biomedical Research Training (CBRT) Program at Texas A&M University has been a successful interdisciplinary program for over a decade. The program continues to be refined in response to the national need for Doctors of Veterinary Medicine (D.V.M/V.M.D) with expertise in cutting-edge biomedical research skills. The mission of the CBRT is to provide trainees with expertise that prepares them to adapt to emerging technologies to solve biomedical and health-related problems. In addition, our program places paramount importance in enabling its trainees to become successful principal investigators in the biomedical research field, able to secure substantial funding to forward their research. The group of research mentors include more than 30 participating faculty across the School of Veterinary Medicine & Biomedical Sciences (VMBS), School of Medicine, School of Public Health, College of Agriculture and Life Sciences, and College of Arts and Sciences. The prospective CBRT trainees/mentees are offered a broad range of research options under a high-level mentor support framework offering high outstanding quality training in basic biomedical research.
Contact Information
T32 Comparative Biomedical Research Training for Veterinarians (CBRT)
Texas A&M University
School of Veterinary Medicine and Biomedical Sciences
Department of Veterinary Pathobiology (VTPB)
Veterinary & Biomedical Education Complex
660 Raymond Stotzer Pkwy
College Station, TX 77843-4474
Grant Number: T32OD011083
Program Director
Dr. Albert Mulenga, B.V.M. (D.V.M.), M.V.Sc., Ph.D.
Professor and Interim Department Head
Fulbright Fellow | Presidential Impact Fellow
Department of Veterinary Pathobiology (VTPB)
Phone: 979-862-2924
Lab office: 979-458-4300
Fax: 979-458-0321
[email protected]; [email protected]
Virginia Polytechnic Institute and State University, Blacksburg, Virginia
Major Areas of Interest

The NIH T32 post-D.V.M. Animal Model Research for Veterinarians (AMRV) training program at the Virginia-Maryland College of Veterinary Medicine at Virginia Tech provides structured in-depth training to veterinarians in the areas of animal models of human diseases in the fields of immunology and inflammation, infectious diseases, neurobiology, public health, cardiology, and regenerative medicine. Trainees will enroll in the Biomedical and Veterinary Sciences graduate program that will expose them to state-of-the-art research skills leading to a Ph.D. degree and challenge them to become independent problem-solvers. After completion of the training program, trainees are expected to assume leadership roles related to the nation's biomedical research agenda in academia, government, and industry.
Contact Information
Virginia-Maryland College of Veterinary Medicine
Virginia Polytechnic Institute and State University
Blacksburg, VA 24061-0042
Grant Number: T32OD028239
Program Director
Xiang-Jin Meng, M.D., Ph.D.
Phone: 540-231-6912
[email protected]
University of Wisconsin School of Veterinary Medicine
Major Areas of Interest

The University of Wisconsin–Madison School of Veterinary Medicine, Comparative Biomedical Sciences Ph.D. program offers cutting-edge research training to graduate veterinarians in infectious diseases (bacteriology/immunology/parasitology/virology), developmental biology, oncology, regenerative medicine, neuroscience, and cellular and molecular physiology.
Contact Information
Grant Number: T32OD010423-16A1
Training Grant Director
Marulasiddappa Suresh, D.V.M., Ph.D.
[email protected]
Program Administrator
Jenny Dahlberg
[email protected]
Program Director
Marulasiddappa Suresh
Phone: 608-265-9791
Fax: 608-890-1774
[email protected]
University of California, Davis
Major Areas of Interest
The University of California, Davis (UC Davis) Veterinary Student Research Training Program provides summer research training to veterinary students. The overall objective of the program is to place interested students with mentors in active laboratories that are engaged in biomedical research. Students are fully immersed in NIH-funded laboratories to achieve experience in research and to gain an appreciation for the importance of research to the veterinary profession. The Program includes faculty from both the School of Medicine and School of Veterinary Medicine who are engaged in research with major areas of emphasis in infectious diseases, cancer, and genomics. The Program interfaces with the campus laboratory animal medicine program, the adjacent California National Primate Research Center, and residency programs in veterinary pathology and laboratory animal medicine. Students are placed with mentors in various programs in the School of Medicine or School of Veterinary Medicine, allowing customization of training emphasis to the students' interests. In addition to research exposure, students participate in seminars on ethics of animal-related research and safety and symposia in which they present their research progress.
Contact Information
University of California, Davis
School of Medicine and School of Veterinary Medicine
One Shields Avenue
https://www.vetmed.ucdavis.edu/research/student-research/star
Grant Number: T35OD010956
Principal Investigator
Danika Bannasch, D.V.M., Ph.D.
Phone: 530-754-8728
[email protected]
Georgia
Major Areas of Interest

The aim of the Georgia Veterinary Scholars Research Program (GVSP) at the University of Georgia College of Veterinary Medicine (UGA-CVM) is to immerse academically talented veterinary students from across the United States in cutting-edge research, inspiring them toward careers in biomedical research and fostering in them a deeper understanding of the goals and needs for veterinarians in research.
Contact Information
Grant Number: T32/T35OD010433
Program Director
Susan Sanchez, Ph.D., FRSB
Phone: 706-542-4200
[email protected]
University of Illinois
Major Areas of Interest

The focus of this training program is translational biomedical research, broadly encompassing the research areas of infectious diseases, reproductive biology, epidemiology, neuroscience, oncology, toxicology, nutrition, and behavior. The goal of this Summer Research Training Program is to identify and facilitate the career progression of veterinary students who have the ability and motivation to become a veterinarian scientist. Veterinary students are matched to one of 40 research laboratories that investigate a broad range of biomedical research topics. In collaboration with the faculty mentor, trainees formulate a testable hypothesis, design the experiments, collect and analyze the experimental data, and report the conclusions. Extensive instruction in the Responsible Conduct of Research is provided through orientation week activities and a seminar series.
Contact Information
University of Illinois Urbana-Champaign
College of Veterinary Medicine
2001 Lincoln Avenue, 2522 VWBSB
Urbana, IL 61802
Grant Number: T35OD011145
Principal Investigator
Megan Mahoney, Ph.D.
Phone: 217-333-7578
[email protected]
Iowa State University
Major Areas of Interest
The Iowa State University Veterinary Summer Scholar Program is designed to introduce veterinary medicine students to research in a wide array of areas, such as clinical medicine, animal science, public health, epidemiology, pharmacology, biomechanics, neuroscience, immunology, parasitology, molecular genetics, and cellular or molecular pathology.
Contact Information
Grant Number: T35OD027967
Program Director
Jodi McGill, Ph.D.
Phone: 515-294-3647
[email protected]
Kansas State University
Major Areas of Interest

The Veterinary Research Scholars Program (VRSP) at Kansas State University (KSU) is a 3‑month summer research experience where veterinary students are paired with faculty researchers to conduct summer research. The goal of the VRSP is to introduce veterinary students to biomedical research through an engaging summer experience and to encourage these talented veterinary professional students to enter veterinary careers involving research. There are many excellent faculty researchers who mentor scholars in this program, both within the College of Veterinary Medicine and across the KSU campus and local U.S. Department of Agriculture (USDA) National Bio and Agro-Defense Facility (NBAF) facilities. Faculty mentors associated with the VRSP are both clinical and basic scientists and have overlapping strengths providing scholars experience in four areas of research excellence: (1) epithelial function in health and disease, (2) infection–immunity–public health, (3) animal models–translational research, and (4) basic and applied pharmacology. Throughout the summer, scholars work with their mentors day-to-day to learn laboratory techniques and various procedures for data collection and participate in weekly journal clubs and interactive workshops to learn about research ethics, how to present research findings, and veterinary career opportunities involving research. Scholars also meet veterinary researchers from corporate and government settings and tour various facilities to learn about the many opportunities available to veterinarians. At the end of the summer, scholars present their research as a poster at our KSU Summer Research Poster Session and at the National Veterinary Research Symposium.
Contact Information
Kansas State University
College of Veterinary Medicine
1800 Denison Ave
Manhattan, KS 66506
www.vet.k-state.edu/research/student-opportunities/vet-scholar
Grant Number: T35OD010979
Program Directors
Bruce Schultz, Ph.D.
Phone: 785-532-4839
Fax: 785-532-4557
[email protected]
Kate KuKanich, D.V.M., Ph.D., DACVIM (SAIM)
Phone: 785-532-4282
[email protected]
Michigan State University
Major Areas of Interest
The training program at Michigan State University is designed to provide short-term, mentored research training to students at the beginning of their veterinary training and inspire them for careers in biomedical and translational research. Veterinarians are uniquely qualified to advance health of humans, animals, and the environment via their in-depth training and knowledge in (1) population systems, food safety, and security; (2) recognition and control of infectious diseases that are transmitted among animals and humans (interspecies); and (3) spontaneous animal models of human diseases. With the additional research training that will be acquired through this program, these future veterinary-scientists will be primed to pursue scientific interrogations to address the gaps in knowledge in the animal-human-ecosystem interface and various public health challenges facing the nation and make significant contributions throughout their careers.
Key words: One Health, comparative medicine, spontaneous animal models, infectious disease, antimicrobial resistance, vector biology, microbial pathogenesis, vaccines, novel diagnostics
Contact Information
College of Veterinary Medicine
Michigan State University
784 Wilson Road, G300
East Lansing, MI 48824
Phone: 571-353-3145
Grant Number: 2T35OD016477-21A1
Program Director
Srinand Sreevatsan, M.V.Sc., M.P.H., Ph.D.
Professor, Associate Dean of Research and Graduate Studies Phone: 571-353-3745
[email protected]
Mississippi State University
Major Areas of Interest
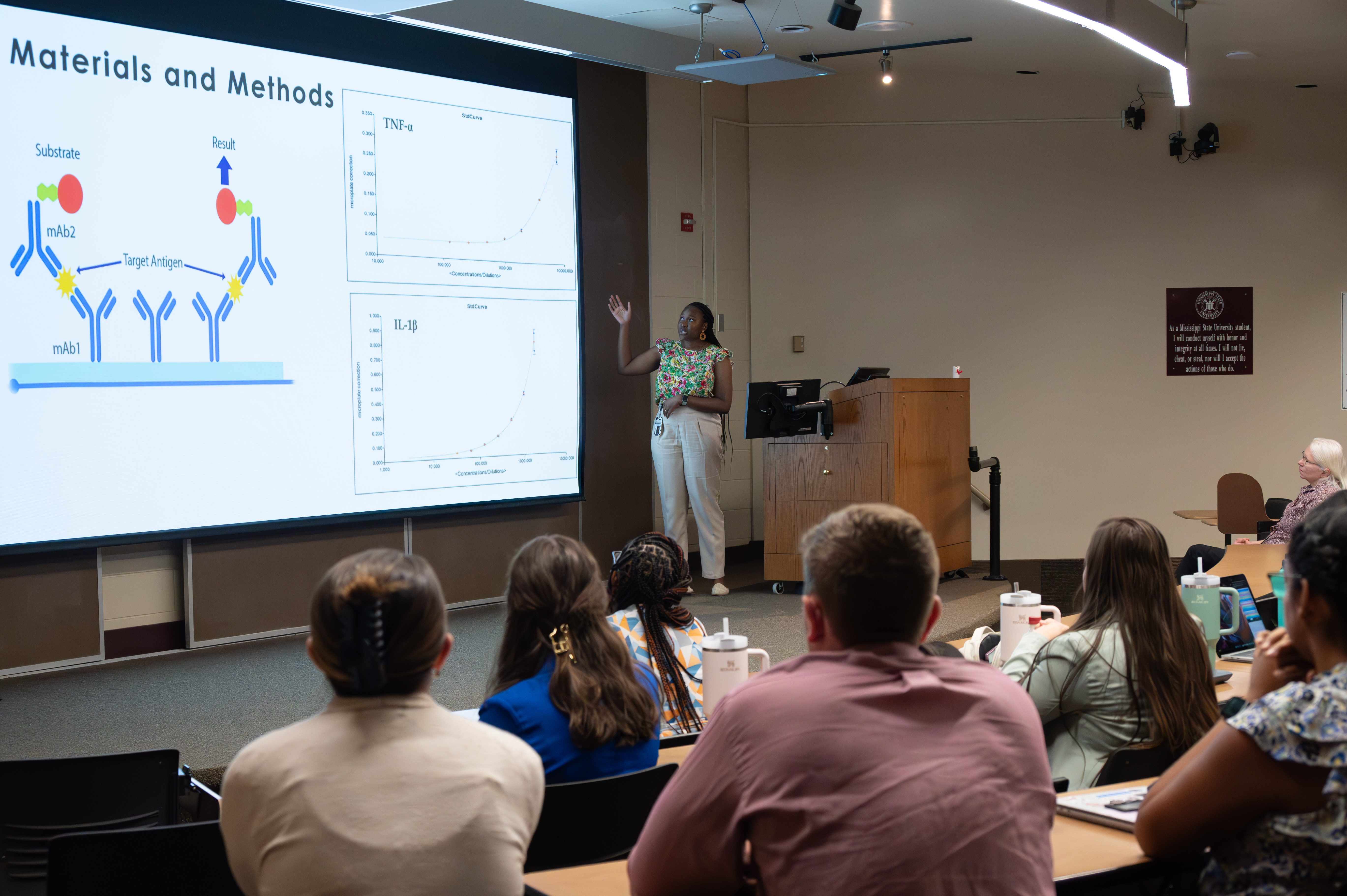
Toxicology/biomedical research (including animal models for human disease)
Population medicine (including antimicrobial stewardship)
Infectious diseases (bacteriology, immunology, parasitology, and virology)
The Summer Research Experience at Mississippi State University College of Veterinary Medicine is called the Vet Med Research Scholars (VMRS) program. The VMRS program is a 12-week investigative program designed to provide veterinary students with training in biomedical research. It provides skills that not only encourage pursuit of research careers but help students develop into leaders within their veterinary profession. Our program is run in conjunction with the Tuskegee University College of Veterinary Medicine, providing 9 positions at Mississippi State and 3 positions at Tuskegee. Major areas of interest within the Tuskegee Veterinary Scholars Program's include One Health, veterinary medicine, and biomedical research.
Contact Information
Grant Number: T35OD010432
Principal Director, Mississippi State University
Barbara Kaplan, Ph.D.
Phone: 662-325-1113
[email protected]
Principal Director, Tuskegee
Temesgen Samuel, D.V.M., Ph.D.
Phone: 334-724-4547
[email protected]
Cornell University, College of Veterinary Medicine
Major Areas of Interest

The Cornell Graduate Training Program in Comparative Medicine combines the very best that Cornell offers in the form of didactic graduate-level instruction, faculty supervision, and training-related activities. Trainees can follow one of two tracks: one is geared to a career in basic research and a second to a career in translational science. Training is structured to ensure the orderly progression of scholars to independence. Research areas available to trainees are broad and include infectious disease, immunology, epidemiology, cancer biology, cell biology and signal transduction, genomics and genetics, developmental biology, molecular medicine, biomedical engineering, and neuroscience.
The program combines independent, faculty-guided research with formal didactic instruction in cell and molecular biology, genomics, and biostatistics; career counseling; and a variety of professional enrichment activities calculated to develop the trainees’ critical capacity, communication, and teamwork skills. Graduate scholars would earn the Ph.D. degree. The average time to degree for veterinarians seeking a Ph.D. at Cornell University is 4.3 years. A Graduate Research Assistantship provided by the College of Veterinary Medicine at Cornell provides stipend to all scholars during the first 9 months of the Ph.D. while trainees are completing three laboratory rotations.
Almost all program alumni go on to obtain faculty positions or positions in industry or government. The mission of our program is to train veterinary scientists that can meet the national need for trained veterinarians within academia, industry, public health, and government to address old and new problems relating to animal and human health.
Contact Information
Grant Number: T35OD010946
Program Directors
John S. Parker, B.V.M.S., Ph.D.; Robert Weiss, Ph.D.
[email protected]
Wake Forest University
Major Areas of Interest
Nonhuman primate medicine, laboratory animal medicine, comparative pathology, anatomic pathology, regenerative medicine, gerosciences, neurosciences and addiction, Alzheimer’s disease, cardiovascular disease, and cancer
The Summer Veterinary Student Research Fellowship at Wake Forest University provides a unique biomedical research experience. Our small program is one of the few veterinary research fellowships embedded in a research-intensive medical hospital. We have our students affiliated with the Department of Pathology, which has a research section on comparative medicine, that is enriched with D.V.M.-faculty and the home for laboratory animal and pathology residencies. Students will work with faculty to create and complete a research proposal over the summer, learning various laboratory, advanced imaging, and pathological techniques. Project are enriched with the study of animal models of human disease, predominantly nonhuman primates but also pigs, rodents, and other laboratory species.
Contact Information
Grant Number: T35OD010946
Program Director
Kylie Kavanagh, D.V.M., M.S., M.P.H.
Professor of Pathology/Comparative Medicine
Phone: 336-713-1475
Fax: 336-716-1515
[email protected]
The Ohio State University
Major Areas of Interest
The training program at The Ohio State University provides short-term intensive research experiences to professional veterinary medical students. The program is based upon a research-intensive summer experience that is part of a larger Summer Research Program. First- and second-year veterinary students select mentors from graduate faculty with primary appointments in Ohio State’s Colleges of Veterinary Medicine (CVM); Medicine; Public Health; and Food, Agricultural, and Environmental Sciences. Projects are developed in collaboration between student and mentor, assuring that the project reflects student interests and ideas. Development of the proposal and preliminary training activities occur prior to summer, and presentation and publication of research results are emphasized in the period following summer. The latter includes an end-of-summer research symposium, during which students make oral presentations of research results and poster/platform presentations at the CVM Research Day the following spring. The faculty-mentor-to-student ratio is 3.3-to-1, providing many training options that ensures a fit with student interests. Summer seminars emphasize science communication, commercialization and intellectual property, and career development. A journal club reinforces concepts in scientific rigor and reproducibility. Topics reflect the breadth of summer projects, and individual sessions include moderation of student presentations by a faculty subject matter expert. A measure of success is a high percentage of student projects leading to publications in peer-reviewed scientific journals.
Contact Information
The Ohio State University College of Veterinary Medicine
1900 Coffey Road
Columbus, OH 43210
https://vet.osu.edu/research/programs/veterinary-student-research-opportunities
Grant Number: T35OD010977
Program Directors
Michael Oglesbee, D.V.M., Ph.D.
Phone: 614-668-1189
[email protected]
Ian Davis, D.V.M., Ph.D.
Phone: 614-292-2954
[email protected]
University of Pennsylvania
Major Areas of Interest

The Penn Vet Short-Term Research Training Program provides research opportunities during the summer months. Students are exposed to multiple areas of biomedical research, including genetics, reproduction, pathogenesis, neurobiology, and cellular and molecular biology. Students gain experience in all aspects of biomedical research including grant writing, bench research, manuscript preparation, poster presentations, and PowerPoint presentations.
Contact Information
University of Pennsylvania
School of Veterinary Medicine
3800 Spruce Street
Philadelphia, PA 19104
Grant Number: T35OD010919
Principal Investigator
Michael Atchison, Ph.D.
Phone: 215-898-6428
[email protected]
Texas A&M University
Major Areas of Interest
The summer Texas A&M University Veterinary Medical Scientist Research Training Program (VMSRTP) provides first- and second-year veterinary students with a robust summer biomedical research experience with experienced NIH-funded mentors and enhanced training in responsible conduct of research methods and scientific communication skills that will enhance their individual research successes and further establish their interest and commitment to veterinary careers in biomedical research. These goals are accomplished through (1) biomedical research in a mentor’s laboratory; (2) at least 8 hours formal training in responsible conduct of research; (3) weekly lunchtime (hour-long) training sessions in scientific communication via oral and poster presentations, abstract and manuscript preparation, as well as training in critical evaluation and presentation of primary journal articles; (4) oral research presentations in the annual Texas A&M VMSRTP Research Symposium; and (5) research poster presentations at the National Veterinary Scholars Symposium. Major areas of research interest by participating faculty mentors within the School of Veterinary Medicine and Biomedical Sciences (as well as the Colleges of Medicine, Science and Agriculture, and Life Sciences) span basic cellular and molecular bench research to translational and clinical research in cancer cell signaling, genetics, and genomics; disease epidemiology and pathogen-host interactions; infectious and vector-borne disease pathogenesis and vaccine development; mechanisms in antibiotic resistance; stem cell biology and regenerative medicine; translational models of musculoskeletal, neurological, and cardiovascular disease; and reproductive and environmental toxicology.
Contact Information
Texas A&M University
School of Veterinary Medicine and Biomedical Sciences
4458 TAMU
College Station, TX 77843-4461
Grant Number: T35OD010991
Principal Investigator
Dana Gaddy, Ph.D.
Phone: 979-862-9134
Fax: 979-847-8981
[email protected]
University of Wisconsin
Major Areas of Interest

The School of Veterinary Medicine provides an opportunity for current first, second, and third-year veterinary medicine students to work with mentors on a wide variety of research projects. Areas of interest include infectious diseases, mouse models of disease, developmental biology, neurosciences, ophthalmology, oncology, orthopedics, pharmacology, stem cell research, reproductive biology, and transplantation.
Contact Information
University of Wisconsin
Department of Surgical Sciences
School of Veterinary Medicine
2015 Linden Drive
Madison, WI 53706
https://www.vetmed.wisc.edu/research/dvm-student-research-experience/summer-scholars-program/
Grant Number: T35OD011078
Principal Investigator
Joan S. Jorgensen, D.V.M., Ph.D.
Phone: 608-890-2337
[email protected]
ORIP Funds Research to Advance Large Animal Models Using Stem Cell Transplantation
Many biomedical researchers use animal models to better understand diseases that occur in humans. Transplantation of testis stem cells—the foundation of sperm production and male fertility—is an established approach developed in mice, but its application to large animals (e.g., sheep, pigs, nonhuman primates) has been more challenging.
Chimpanzee Management Reports
NIH conducts an annual census of its owned and supported chimpanzees at the end of each fiscal year (September 30) to provide the public with official figures on their numbers and cost of care. Due to natural attrition, the total number of chimpanzees is expected to decrease each year. For fiscal year 2025 (FY25), the overall mortality rate was 6.4%, which is within the expected range. The number of chimpanzees at different facilities may also increase or decrease because of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance.
Facility Construction Costs
The federally supported chimpanzee population has been steadily declining since the late 1990s because of a continuing breeding moratorium and natural mortality. This decline in population has led to an increase in unused space at NIH-supported facilities. Although the maintenance of these facilities remains necessary, federal funds are not needed for new construction for these nonhuman primates. As a result, no construction or renovation costs were incurred during FY25.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities—including both sanctuary and non-sanctuary sites—comply with local, state, and federal regulations governing animal research and are certified by AAALAC International. These facilities provide care that exceeds legal requirements, and NIH is committed to maintaining high standards of care, safety, and welfare for all chimpanzees in its supported facilities.
Because the number of chimpanzees at each facility can change over time, the daily cost per animal also varies throughout the year and from year to year. Costs differ among facilities because of such factors as local economic conditions, the size of the chimpanzee population, and whether the facility also houses other nonhuman primates, allowing for shared veterinary expertise and care costs. Generally, a larger population of chimpanzees and other nonhuman primates results in lower per-animal daily costs due to economies of scale. However, as the chimpanzee population continues to decline in the absence of breeding, achieving these economies of scale becomes increasingly difficult. To mitigate this, facilities may adjust staffing levels in proportion to the population decrease, although this adjustment will vary by facility. Relocation of animals to the Federal Sanctuary facility operated by Chimp Haven is ongoing, as medically permissible, to optimize cost efficiency at Chimp Haven while reducing efficiency at other facilities. These relocation efforts will continue in future years.
Based on the census as of October 1, 2025, and the most recent FY25 awards and payments, NIH spent a total of $7.46 million (M) to care for 321 chimpanzees across four facilities, averaging $63.67 per day per animal. The facilities include (1) the Keeling Center for Comparative Medicine and Research (KCCMR) at The University of Texas MD Anderson Cancer Center in Bastrop, Texas; (2) the Southwest National Primate Research Center (SNPRC) at the Texas Biomedical Research Institute in San Antonio, Texas; (3) the Alamogordo Primate Facility (APF) at the Holloman Air Force Base in Alamogordo, New Mexico; and (4) the Federal Sanctuary facility operated by Chimp Haven in Keithville, Louisiana.
TABLE: FY25 Funds Provided for NIH-Owned and NIH-Supported Chimpanzees* as of October 1, 2025
| Facility | Total Number of Chimpanzees | Total NIH Cost**, $M/Year | Average NIH Cost***, $/Animal/Day | Number of Chimpanzees Not to Be Relocated**** |
|---|---|---|---|---|
| KCCMR | 37 | $1.37 | $101.44 | 37a |
| SNPRC | 7 | $0.00 | $0.00b | 7 |
| APF | 0c | $0.14 | Not Applicabled | Not Applicable |
| Chimp Haven | 277 | $5.95 | $58.85e | Not Applicable |
| Total |
321 |
$7.46 |
Overall $63.67 |
Not Applicable |
*Definition of NIH-owned and -supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care.
- NIH-supported: Chimpanzees are privately owned, and NIH funded their cost of care through FY20.
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals, and thus the costs per animal per day, will vary throughout the year at all facilities.
****Determined by the NIH Veterinary Panel (https://dpcpsi.nih.gov/council/chimpanzee_advice).
aThere are six chimpanzees located at KCCMR that are not being moved at this time because they serve as behavioral support for at-risk chimpanzees that are not being relocated from KCCMR based on the recommendation of the NIH Veterinary Panel. Each of these six KCCMR chimpanzees will be reevaluated for potential transfer to Chimp Haven based on age, health, behavior, social grouping, and environmental needs when the animal or animals that they support die.
bNo NIH funding was provided in FY25 for support for these animals, as explained below under “Current Costs Per Facility.”
cIn November 2024, NIH decided to relocate the remaining chimpanzees from APF to Chimp Haven. The relocation of these animals was completed in March 2025.
dNIH funding for FY25 support of the animals at APF from October 2024 to March 2025 is explained below under “Current Costs Per Facility.”
eNIH funding for FY25 provides only partial support for these animals as per the Chimpanzee Health Improvement, Maintenance, and Protection (CHIMP) Act.
Cost Determinations
Total Costs Per Year for the NIH-Owned or -Supported Chimpanzees
October 1, 2025, report
- Total number of animals owned and supported = 321
- Total cost for FY25 = $7.46 M
- Total cost per animal per day = $63.67
October 1, 2024, report
- Total number of animals owned and supported = 343
- Total cost for FY24 = $10.99 M
- Total cost per animal per day = $87.78
October 1, 2023, report
- Total number of animals owned and supported = 371
- Total cost for FY23 = $8.52 M
- Total cost per animal per day = $62.92
October 1, 2022, report
- Total number of animals owned and supported = 390
- Total cost for FY22 = $8.3 M
- Total cost per animal per day = $58.31
October 1, 2021, report
- Total number of animals owned and supported = 422
- Total cost for FY21 = $6.38 M
- Total cost per animal per day = $41.42
October 1, 2020, report
- Total number of animals owned and supported = 436
- Total cost for FY20 = $6.16 M
- Total cost per animal per day = $38.71
October 18, 2019, report
- Total number of animals owned and supported = 463
- Total cost for FY19 = $7.59 M
- Total cost per animal per day = $44.91
October 1, 2018, report
- Total number of animals owned and supported = 494
- Total cost for FY18 = $8.72 M
- Total cost per animal per day = $48.36
October 1, 2017, report
- Total number of animals owned and supported = 518
- Total cost for FY17 = $9.62 M
- Total cost per animal per day = $50.88
October 24, 2016, report
- Total number of animals owned and supported = 542
- Total cost for FY16 = $9.06 M
- Total cost per animal per day = $45.80
October 21, 2015, report
- Total number of animals owned and supported = 573
- Total cost for FY15 = $10.19 M
- Total cost per animal per day = $48.77
October 27, 2014, report
- Total number of animals owned and supported = 606
- Total cost for FY14 = $12.09 M
- Total cost per animal per day = $54.66
October 27, 2013, report
- Total number of animals owned and supported = 644
- Total cost for FY13 = $9.76 M
- Total cost per animal per day = $41.50
October 23, 2012, report
- Total number of animals owned and supported = 669
- Total cost for FY12 = $12.38 M
- Total cost per animal per day = $50.60
October 31, 2011, report
- Total number of animals owned and supported = 713
- Total cost for FY11 = $11.58 M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR—Chimpanzee Care Center, Bastrop, Texas, operated by the Keeling Center for Comparative Medicine and Research as part of The University of Texas MD Anderson Cancer Center, under contract with NIH.
- The total cost to NIH for FY25 under the contract with The University of Texas MD Anderson Cancer Center is $1,368,250.
SNPRC (P51)—Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, Texas.
- Under the previous P51 award project period (05/18/2016–04/30/2021), costs for care were shared among the chimpanzees and several thousand other nonhuman primates.
- No funds were obligated in FY25, as costs for the care of the chimpanzees were not included in the current P51 award project period (05/15/2021–04/30/2026).
- Since May 1, 2021, the chimpanzees owned by the SNPRC have been supported with funds provided solely by the Texas Biomedical Research Institute.
APF—Alamogordo Primate Facility, Alamogordo, New Mexico, operated by Charles River Laboratories (a for-profit company), under contract with NIH.
- The total cost to NIH for FY25 to support APF is $142,712.
- $142,712 was obligated in FY25 to cover costs incurred during the remaining 6 months of the base year of a new contract (02/01/2025–07/30/2025).
- NIH did not exercise option year 1, thus ending the contract.
Chimp Haven, Inc.—A not-for-profit company that operates the Federal Sanctuary in Keithville, Louisiana, under contract with NIH.
- The total cost to NIH for FY25 to support the Federal Sanctuary is $5,950,680.
- $950,680 was obligated in FY25 to cover costs incurred during the remaining 6 months of option year 2 (02/01/2025–07/31/2025).
- $5 M initially was obligated in FY25 to cover projected costs for the first 6 months of option year 3 (08/01/2025–01/31/2026), with future costs for the remaining 6 months for this option year (02/01/2026–07/31/2026) and any additional costs incurred during the first 6 months of the next option year (08/01/2026–01/31/2027) to be covered by FY26 funds.
- NIH funding for the care of chimpanzees at the Federal Sanctuary is made through a contract with matching requirements—the contractor (Chimp Haven, Inc.) must match $1 for every $3 from NIH for operating expenses.
NIH conducts an annual census of its owned and supported chimpanzees following the end of each fiscal year (September 30) to provide the public with official figures on their numbers and cost of care. Due to natural attrition, the total number of chimpanzees is expected to decrease each year. For Fiscal Year 2024 (FY24), the overall mortality rate was 7.5%, which is within the expected range. The number of chimpanzees at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction; and (2) chimpanzee care and facility maintenance.
Facility Construction Costs
The federally supported chimpanzee population has been steadily declining since the late 1990s due to a continuing breeding moratorium and natural mortality. This decline in population has led to an increase in unused space at NIH-supported facilities. While the maintenance of these facilities remains necessary, there is no need for federal funds for new construction for these nonhuman primates. As a result, there were no construction or renovation costs incurred during FY24.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities, including both sanctuary and non-sanctuary sites, comply with local, state, and federal regulations governing animal research. Additionally, they are certified by the Association for Assessment and Accreditation of Laboratory Animal Care, International. These facilities provide care that exceeds legal requirements, and NIH is committed to maintaining high standards of care, safety, and welfare for all chimpanzees in its supported facilities.
Since the number of chimpanzees at each facility can change over time, the daily cost per animal also varies throughout the year and from year to year. Costs differ among facilities due to factors such as local economic conditions, size of the chimpanzee population, and whether the facility also houses other nonhuman primates, which allows for shared veterinary expertise and care costs. Generally, a larger population of chimpanzees and other nonhuman primates results in lower per-animal daily costs due to economies of scale. However, as the chimpanzee population continues to decline in the absence of breeding, achieving these economies of scale becomes increasingly difficult. To mitigate this, facilities may adjust staffing levels in proportion to the population decrease, though this adjustment will vary by facility. Relocation of animals to the Federal Sanctuary at Chimp Haven is ongoing, as medically permissible, to optimize cost efficiency at Chimp Haven while reducing efficiency at other facilities. These relocation efforts will continue in future years.
Based on the census as of October 1, 2024, and the most recent FY24 awards and payments, NIH spent a total of $10.99M to care for 343 chimpanzees across four facilities, averaging $87.78 per day per animal. The facilities include: 1) the Keeling Center for Comparative Medicine and Research (KCCMR) at the University of Texas MD Anderson Cancer Center in Bastrop, Texas; 2) the Southwest National Primate Research Center (SNPRC) at the Texas Biomedical Research Institute in San Antonio, Texas; 3) the Alamogordo Primate Facility (APF) at the Holloman Air Force Base in Alamogordo, New Mexico; and 4) the Federal Sanctuary facility operated by Chimp Haven, Inc. in Keithville, Louisiana.
TABLE: FY24 Funds Provided for NIH-Owned and NIH-Supported Chimpanzees* as of October 1, 2024
| Facility | Total # of chimpanzees | Total NIH Cost**, $M/year | Average NIH Cost***, $/animal/day | # of chimpanzees not to be relocated |
|---|---|---|---|---|
| KCCMR | 41 | 1.48 | $98.90 | 41 |
| SNPRC | 7 | 0.00 | $0.00x | 7 |
| APF | 23 | 1.93 | $229.90xx | 23 |
| Chimp Haven | 272 | 7.58 | $76.35xxx | Not Applicable |
| Total |
343 |
10.99 |
Overall $87.78 |
Not Applicable |
*Definition of NIH-owned and -supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned, and NIH funded their cost of care through FY20.
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
****Determined by the NIH Veterinary Panel (https://dpcpsi.nih.gov/council/chimpanzee_advice). There are 6 chimpanzees located at KCCMR that are not being moved at this time because they serve as behavioral support for at risk chimpanzees that are not being relocated from KCCMR based on the recommendation of the NIH Veterinary Panel. Each of these 6 KCCMR chimpanzees will be re-evaluated for potential transfer to Chimp Haven based on age, health, behavior, social grouping, and environmental needs when the animal or animals being supported by them dies.
×No NIH funding was provided in FY24 for support for these animals as explained below under “Current Costs Per Facility”.
××NIH funding for FY24 support of the animals at APF is explained below under “Current Costs Per Facility” (calculated for the periods 02/01/2024 – 07/30/2024, 07/31/2024 – 09/25/2024 and 09/26/2024 - 01/31/2025).
×××NIH funding for FY24 provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH-Owned or -Supported Chimpanzees
October 1, 2024 report
- Total number of animals owned and supported = 343
- Total cost for FY24 = $10.99M
- Total cost per animal per day = $87.78
October 1, 2023 report
- Total number of animals owned and supported = 371
- Total cost for FY23 = $8.52M
- Total cost per animal per day = $62.92
October 1, 2022 report
- Total number of animals owned and supported = 390
- Total cost for FY22 = $8.3M
- Total cost per animal per day = $58.31
October 1, 2021 report
- Total number of animals owned and supported = 422
- Total cost for FY21 = $6.38M
- Total cost per animal per day = $41.42
October 1, 2020 report
- Total number of animals owned and supported = 436
- Total cost for FY20 = $6.16M
- Total cost per animal per day = $38.71
October 18, 2019 report
- Total number of animals owned and supported = 463
- Total cost for FY19 = $7.59M
- Total cost per animal per day = $44.91
October 1, 2018 report
- Total number of animals owned and supported = 494
- Total cost for FY18 = $8.72M
- Total cost per animal per day = $48.36
October 1, 2017 report
- Total number of animals owned and supported = 518
- Total cost for FY17 = $9.62M
- Total cost per animal per day = $50.88
October 24, 2016 report
- Total number of animals owned and supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 21, 2015 report
- Total number of animals owned and supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 27, 2014 report:
- Total number animals owned and supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 27, 2013 report:
- Total number of animals owned and supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 23, 2012 report:
- Total number of animals owned and supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 31, 2011 report:
- Total number of animals owned and supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR – Chimpanzee Care Center, Bastrop, TX is operated by the Keeling Center for Comparative Medicine and Research as part of the University of Texas MD Anderson Cancer Center under contract with the NIH.
- The total cost to NIH for FY24 under the contract with the University of Texas MD Anderson Cancer Center is $1,481,969.
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Under the previous P51 award project period (05/18/2016 - 04/30/2021), costs were shared between care for the chimpanzees and several thousand other non-human primates.
- No funds were obligated in FY24 as costs for the care of the chimpanzees were not included in the current P51 award project period (05/15/2021 – 04/30/2026).
- After 05/01/2021, the chimpanzees owned by the SNPRC are supported with funds provided solely by the Texas Biomedical Research Institute.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- The total cost to NIH for FY24 to support APF is $1,925,548.
- $709,969 was obligated in FY24 to cover costs incurred during the remaining 6 months of option year four (02/01/2024 – 07/30/2024).
- $215,579 was obligated in FY24 to cover costs incurred during an extension exercised at the end of option year four (07/31/2024 to 09/25/2024).
- $1,000,000 was obligated in FY24 to initially cover projected costs for the first 4.5 months of the base year of a new contract (09/26/2024 – 01/31/2025), with future costs for the remaining months for this base year (02/01/2025 – 07/30/2025) and any additional costs incurred during the first 6 months of option year one (07/31/2025 – 01/31/2026) to be covered by FY25 funds.
Chimp Haven, Inc. - A not-for-profit company that operates the Federal Sanctuary in Keithville, LA under contract with the NIH.
- The total cost to NIH for FY24 to support the Federal Sanctuary is $7,583,310.
- $2,631,178 was obligated in FY24 to cover costs incurred during the remaining 6 months of option year one (02/01/2024 – 07/31/2024).
- $4,952,132 was obligated in FY24 to initially cover projected costs for the first 6 months of option year two (08/01/2024 - 01/31/2025), with future costs for the remaining 6 months for this option year (02/01/2025 – 07/31/2025) and any additional costs incurred during the first 6 months of the next option year (08/01/2025 – 01/31/2026) to be covered by FY25 funds.
- NIH funding for the care of chimpanzees at the Federal Sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 NIH dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 1, 2023
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (September 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of natural attrition. The overall mortality rate for Fiscal Year 2023 (FY23) was 4.9%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction; and (2) chimpanzee care and facility maintenance costs.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A decrease in census results in an increase in unused space at all of the NIH-supported facilities. While the need for maintenance continues, there is no need for new construction of facilities for these nonhuman primates. There were no construction or renovation costs during FY23.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state, and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International. These facilities provide care that exceeds that required by law. NIH is committed to continuing to provide high quality care and ensuring the safety and welfare of the chimpanzees at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility will vary over the course of a year and from year to year. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates, where expensive care and veterinary expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the costs per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a specific facility. This can be partially compensated by decreasing staff by a certain percentage as the chimpanzee population decreases (this will vary between facilities). Animals have been relocated to the Federal Sanctuary at Chimp Haven, so NIH expects the cost efficiency to be optimized at Chimp Haven while the efficiency at other facilities will decrease. Relocation efforts, as medically permissible, will continue into future years.
Based on the census as of October 1, 2023, and the most recent FY23 awards and payments, NIH spent a total of $8.52M for 371 animals for an average of $62.92 per day per chimpanzee across all four of the facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR) at the University of Texas MD Anderson Cancer Center in Bastrop, Texas; 2) the Southwest National Primate Research Center (SNPRC) at the Texas Biomedical Research Institute in San Antonio, Texas; 3) the Alamogordo Primate Facility (APF) at the Holloman Air Force Base in Alamogordo, New Mexico; and 4) the Federal Sanctuary facility operated by Chimp Haven, Inc. in Keithville, Louisiana.
TABLE: FY23 Funds Provided for NIH-Owned and NIH-Supported Chimpanzees* as of October 1, 2023
| Facility | Total # of chimpanzees | Total NIH Cost**, $M/year | Average NIH Cost***, $/animal/day | # of chimpanzees not to be relocated |
|---|---|---|---|---|
| KCCMR | 43 | 1.68 | $107.04 | 43 |
| SNPRC | 10 | 0.00 | $0.00× | 10 |
| APF | 28 | 1.32 | $129.16×× | 28 |
| Chimp Haven | 290 | 5.52 | $52.15××× | Not Applicable |
| Total | 371 | 8.52 |
Overall $62.92 |
Not Applicable |
*Definition of NIH-owned and -supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned, and NIH funded their cost of care through FY20.
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
****Determined by the NIH Veterinary Panel (https://dpcpsi.nih.gov/council/chimpanzee_advice). There are 6 chimpanzees located at KCCMR and 2 chimpanzees at SNPRC that are not being moved at this time because they serve as behavioral support for another chimpanzee that is not being moved. If that paired chimpanzee dies, the other chimpanzee will be re-evaluated for potential transfer to Chimp Haven based on their age, health, behavior, social grouping, and environmental needs.
× No NIH funding was provided in FY23 for support for these animals as explained under “Current Costs Per Facility”.
××The NIH funding for FY23 provides partial support for the animals as explained under “Current Costs Per Facility” ((calculated for the periods 02/01/2023 – 07/30/2023 and 07/31/2023 – 01/31/2024).
×××The NIH funding for FY23 provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH-Owned or -Supported Chimpanzees
October 1, 2023 report
- Total number of animals owned and supported = 371
- Total cost for FY23 = $8.52M
- Total cost per animal per day = $62.92
October 1, 2022 report
- Total number of animals owned and supported = 390
- Total cost for FY22 = $8.3M
- Total cost per animal per day = $58.31
October 1, 2021 report
- Total number of animals owned and supported = 422
- Total cost for FY21 = $6.38M
- Total cost per animal per day = $41.42
October 1, 2020 report
- Total number of animals owned and supported = 436
- Total cost for FY20 = $6.16M
- Total cost per animal per day = $38.71
October 18, 2019 report
- Total number of animals owned and supported = 463
- Total cost for FY19 = $7.59M
- Total cost per animal per day = $44.91
October 1, 2018 report
- Total number of animals owned and supported = 494
- Total cost for FY18 = $8.72M
- Total cost per animal per day = $48.36
October 1, 2017 report
- Total number of animals owned and supported = 518
- Total cost for FY17 = $9.62M
- Total cost per animal per day = $50.88
October 24, 2016 report
- Total number of animals owned and supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 21, 2015 report
- Total number of animals owned and supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 27, 2014 report:
- Total number animals owned and supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 27, 2013 report:
- Total number of animals owned and supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 23, 2012 report:
- Total number of animals owned and supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 31, 2011 report:
- Total number of animals owned and supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR – Chimpanzee Care Center, Bastrop, TX is operated by the Keeling Center for Comparative Medicine and Research as part of the University of Texas MD Anderson Cancer Center under contract with the NIH.
- The total cost to NIH for FY23 under the contract with the University of Texas MD Anderson Cancer Center is $1,680,672 (12/01/2022-11/30/2023).
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Under the previous P51 award project period (05/18/2016 - 04/30/2021), costs were shared between care for the chimpanzees and several thousand other non-human primates.
- No funds were obligated in FY23 as costs for the care of the chimpanzees were not included in the current P51 award project period (05/15/2021 – 04/30/2026).
- After 05/01/2021, the chimpanzees owned by the SNPRC are supported with funds provided solely by the Texas Biomedical Research Institute.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- The total cost to NIH for FY23 under the contract with Charles River Laboratories is $1,321,284.
- $681,284 was obligated in FY23 to cover costs incurred during the remaining 6 months of option year three (02/01/2023 – 07/30/2023).
- $640,000 was obligated in FY23 to initially cover projected costs for the first 6 months of option year four (07/31/2023 - 01/31/2024), with future costs for the remaining 6 months for this option year (02/01/2024 – 07/30/2024) and any additional costs incurred during the first 6 months of the next option year (07/31/2024 – 01/31/2025) to be covered by FY24 funds.
Chimp Haven, Inc. - A not-for-profit company that operates the Federal Sanctuary in Keithville, LA under contract with the NIH.
- The total cost to NIH for FY23 to support the Federal Sanctuary is $5,520,148.
- $2,520,148 was obligated in FY23 to cover costs incurred during the remaining 6 months of the base year (02/01/2023 – 07/31/2023).
- $3,000,000 was obligated in FY23 to initially cover projected costs for the first 6 months of option year one (08/01/2023 - 01/31/2024), with future costs for the remaining 6 months for this option year (02/01/2024 – 07/30/2024) and any additional costs incurred during the first 6 months of the next option year (07/31/2024 – 01/31/2025) to be covered by FY24 funds.
- NIH funding for the care of chimpanzees at the Federal Sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 NIH dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 1, 2022
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (September 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of natural attrition. The overall mortality rate for Fiscal Year 2022 (FY22) was 7.6%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction; and (2) chimpanzee care and facility maintenance costs.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A decrease in census results in an increase in unused space at all of the NIH-supported facilities. While the need for maintenance continues, there is no need for new construction of facilities for these nonhuman primates. There were no construction or renovation costs during FY22.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International. These facilities provide care that exceeds that required by law. NIH is committed to continuing to provide high quality care and ensuring the safety and welfare of the chimpanzees at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility will vary over the course of a year and from year to year. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates, where expensive care and veterinary expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the costs per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a specific facility. This can be partially compensated by decreasing staff by a certain percentage as the chimpanzee population decreases (this will vary between facilities). Currently, animals are being relocated to the Federal Sanctuary at Chimp Haven, so NIH expects to see the cost efficiency begin to optimize at Chimp Haven while the efficiency at other facilities will decrease. Relocation efforts, as medically permissible, will continue into future years.
Based on the census as of October 1, 2022, and the most recent FY22 awards and payments, NIH spent a total of $8.3M for 390 animals for an average of $58.31 per day per chimpanzee across all four of the facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR) at the University of Texas MD Anderson Cancer Center in Bastrop, Texas; 2) the Southwest National Primate Research Center (SNPRC) at the Texas Biomedical Research Institute in San Antonio, Texas; 3) the Alamogordo Primate Facility (APF) at the Holloman Air Force Base in Alamogordo, New Mexico; and 4) the Federal Sanctuary facility operated by Chimp Haven, Inc. in Keithville, Louisiana.
TABLE: FY22 Funds Provided for NIH-Owned and NIH-Supported Chimpanzees* as of October 1, 2022
| Facility | Total # of chimpanzees | Total NIH Cost**, $M/year | Average NIH Cost***, $/animal/day | # of chimpanzees not to be relocated |
|---|---|---|---|---|
| KCCMR | 46 | 1.55 | $92.32 | 46 |
| SNPRC | 10 | 0.00 | $0.00× | 10 |
| APF | 30 | 1.30 | $118.72×× | 30 |
| Chimp Haven | 304 | 5.45 | $49.12××× | Not Applicable |
| Total | 390 | 8.30 |
Overall $58.31 |
Not Applicable |
*Definition of NIH-owned and -supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned, and NIH funded their cost of care through FY20.
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
****Determined by the NIH Veterinary Panel (https://dpcpsi.nih.gov/council/chimpanzee_advice). There are 7 chimpanzees located at KCCMR and 2 chimpanzees at SNPRC that are not being moved at this time because they serve as behavioral support for another chimpanzee that is not being moved. If that paired chimpanzee dies, the other chimpanzee will be re-evaluated for potential transfer to Chimp Haven based on their age, health, behavior, social grouping, and environmental needs.
× No NIH funding was provided in FY22 for support for these animals as explained under “Current Costs Per Facility”.
××The NIH funding for FY22 provides partial support for the animals as explained under “Current Costs Per Facility” (calculated for the periods 02/01/2022 – 07/30/2022 and 07/31/2022 – 01/31/2023).
×××The NIH funding for FY22 provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH-Owned or -Supported Chimpanzees
October 1, 2022 report
- Total number of animals owned and supported = 390
- Total cost for FY22 = $8.3M
- Total cost per animal per day = $58.31
October 1, 2021 report
- Total number of animals owned and supported = 422
- Total cost for FY21 = $6.38M
- Total cost per animal per day = $41.42
October 1, 2020 report
- Total number of animals owned and supported = 436
- Total cost for FY20 = $6.16M
- Total cost per animal per day = $38.71
October 18, 2019 report
- Total number of animals owned and supported = 463
- Total cost for FY19 = $7.59M
- Total cost per animal per day = $44.91
October 1, 2018 report
- Total number of animals owned and supported = 494
- Total cost for FY18 = $8.72M
- Total cost per animal per day = $48.36
October 1, 2017 report
- Total number of animals owned and supported = 518
- Total cost for FY17 = $9.62M
- Total cost per animal per day = $50.88
October 24, 2016 report
- Total number of animals owned and supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 21, 2015 report
- Total number of animals owned and supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 27, 2014 report:
- Total number animals owned and supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 27, 2013 report:
- Total number of animals owned and supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 23, 2012 report:
- Total number of animals owned and supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 31, 2011 report:
- Total number of animals owned and supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR – Chimpanzee Care Center, Bastrop, TX is operated by the Keeling Center for Comparative Medicine and Research as part of the University of Texas MD Anderson Cancer Center under contract with the NIH.
- The total cost to NIH for FY22 under the contract with the University of Texas MD Anderson Cancer Center is $1,550,000 (12/01/2021-11/30/2022).
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Under the previous P51 award project period (05/18/2016 - 04/30/2021), costs were shared between care for the chimpanzees and several thousand other non-human primates.
- No funds were obligated in FY22 as costs for the care of the chimpanzees were not included in the current P51 award project period (05/15/2021 – 04/30/2026).
- After 05/01/2021, the chimpanzees owned by the SNPRC are supported with funds provided solely by the Texas Biomedical Research Institute.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- The total cost to NIH for FY22 under the contract with Charles River Laboratories is $1,304,059.
- $304,059 was obligated in FY22 to cover costs incurred during the remaining 6 months of option year two (02/01/2022 – 07/30/2022).
- $1,000,000 was obligated in FY22 to initially cover projected costs for the first 6 months of option year three (07/31/2022 - 01/31/2023), with future costs for the remaining 6 months for this option year (02/01/2023 – 07/30/2023) and any additional costs incurred during the first 6 months of the next option year (07/31/2023 – 01/31/2024) to be covered by FY23 funds.
Chimp Haven, Inc. - A not-for-profit company that operates the Federal Sanctuary in Keithville, LA under contract with the NIH.
- The total cost to NIH for FY22 to support the Federal Sanctuary is $5,450,000.
- Additional FY22 costs incurred towards the final option year are $2,450,000 (09/30/2021 - 07/31/2022).
- The total cost to NIH for FY22 under the new contract issued to support the Federal Sanctuary is $3,000,000 (08/01/2022 - 07/31/2023).
- NIH funding for the care of chimpanzees at the Federal Sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 NIH dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 1, 2021
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (September 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. The overall mortality rate for Fiscal Year 2021 (FY21) was 3.2%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction; and (2) chimpanzee care and facility maintenance costs.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A decrease in census results in an increase in unused space at all of the NIH-supported facilities. While the need for maintenance still arises, there is no need for new construction of facilities for these nonhuman primates. There were no construction or renovation costs during FY21.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International. These facilities provide care that exceeds that required by law. NIH is committed to maintaining high quality of (or optimum) care and welfare at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility will vary over the course of a year and from year to year. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates, where expensive care and veterinary expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the costs per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a specific facility. This can be partially compensated by decreasing staff by a certain percentage as the chimpanzee population decreases (this will vary between facilities). Currently, animals are being relocated to the Federal Sanctuary at Chimp Haven, so NIH expects to see the cost efficiency begin to optimize at Chimp Haven while the efficiency at other facilities will decrease. Relocation efforts will continue into future years.
Based on the census as of October 1, 2021, and the most recent FY21 awards and payments, NIH spent a total of $6.38M for 422 animals for an average of $41.42 per day per chimpanzee across all four of the facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR) at the University of Texas MD Anderson Cancer Center in Bastrop, Texas; 2) the Southwest National Primate Research Center (SNPRC) at the Texas Biomedical Research Institute in San Antonio, Texas; 3) the Alamogordo Primate Facility (APF) at the Holloman Air Force Base in Alamogordo, New Mexico; and 4) the Federal Sanctuary facility operated by Chimp Haven, Inc. in Keithville, Louisiana.
TABLE: FY21 Funds Provided for NIH-Owned and NIH-Supported Chimpanzees* as of October 1, 2021
|
Facilities |
||||
|---|---|---|---|---|
| Facility | Total # of chimpanzees | Total NIH Cost**, $M/year | Average NIH Cost***, $/animal/day | # of chimpanzees not to be relocated**** |
| KCCMR | 51 | 1.80 | $96.70 | 49 |
| SNPRC | 19 | 0.00 | $0.00× | Under Review |
| APF | 35 | 1.58 | $123.68×× | 35 |
| Chimp Haven | 317 | 3.00 | $25.93××× | Not Applicable |
| Total | 422 | 6.38 |
Overall $41.42 |
Not Applicable |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned, and NIH funded their cost of care through FY20.
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
****Determined by the NIH Veterinary Panel (https://dpcpsi.nih.gov/council/chimpanzee_advice).
× No NIH funding was provided in FY21 for support for these animals as explained under “Current Costs Per Facility”.
××The NIH funding for FY21 provides partial support for the animals as explained under “Current Costs Per Facility” (calculated for the periods 02/01/2021 – 07/30/2021 and 07/31/2021 – 01/31/2022).
×××The NIH funding for FY21 provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH-Owned or -Supported Chimpanzees
October 1, 2021 report
- Total number of animals owned and supported = 422
- Total cost for FY21 = $6.38M
- Total cost per animal per day = $41.42
October 1, 2020 report
- Total number of animals owned and supported = 436
- Total cost for FY20 = $6.16M
- Total cost per animal per day = $38.71
October 18, 2019 report
- Total number of animals owned and supported = 463
- Total cost for FY19 = $7.59M
- Total cost per animal per day = $44.91
October 1, 2018 report
- Total number of animals owned and supported = 494
- Total cost for FY18 = $8.72M
- Total cost per animal per day = $48.36
October 1, 2017 report
- Total number of animals owned and supported = 518
- Total cost for FY17 = $9.62M
- Total cost per animal per day = $50.88
October 24, 2016 report
- Total number of animals owned and supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 21, 2015 report
- Total number of animals owned and supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 27, 2014 report:
- Total number animals owned and supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 27, 2013 report:
- Total number of animals owned and supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 23, 2012 report:
- Total number of animals owned and supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 31, 2011 report:
- Total number of animals owned and supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR – Chimpanzee Care Center, Bastrop, TX is operated by the Keeling Center for Comparative Medicine and Research as part of the University of Texas MD Anderson Cancer Center under contract with the NIH.
- The total cost to NIH for FY21 under the contract with the University of Texas MD Anderson Cancer Center is $1,800,000 for the base year (12/01/2020-11/30/2021).
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Under the previous P51 award project period (05/18/2016 - 04/30/2021), costs were shared between care for the chimpanzees and several thousand other non-human primates.
- No funds were obligated in FY21 as costs for the care of the chimpanzees were not included in the current P51 award project period (05/15/2021 – 04/30/2026).
- After 05/01/2021, the chimpanzees owned by the SNPRC are supported with funds provided solely by the Texas Biomedical Research Institute.
- NIH is actively relocating chimpanzees from this facility to the Federal Sanctuary at Chimp Haven, Inc. where their care is covered as described below, unless relocation would severely or irreversibly accelerate deterioration of the chimpanzee’s physical or behavioral health.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- The total cost to NIH for FY21 under the contract with Charles River Laboratories is $1,578,289.
- $500,000 was obligated in FY21 to cover costs incurred during the remaining 6 months of option year one (02/01/2021 – 07/30/2021).
- $1,078,289 was obligated in FY21 to initially cover projected costs for the first 6 months of option year two (07/31/2021 - 01/31/2022), with future costs for the remaining 6 months for this option year (02/01/2022 – 07/30/2022) and any additional costs incurred during the first 6 months of the next option year (07/31/2022 – 01/31/2023) to be covered by FY22 funds.
Chimp Haven, Inc. - A not-for-profit company that operates the Federal Sanctuary in Keithville, LA under contract with the NIH
- NIH-incurred costs under the contract with Chimp Haven, Inc. for period 09/30/2021- 06/30/2022 is $3,000,000, with any additional costs incurred during the final option year being covered by FY22 funds and through a new contract (to be awarded in July 2022 with FY22 funds pending review and acceptance of a proposal).
- NIH funding for the care of chimpanzees at the Federal Sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 NIH dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 1, 2020
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. For example, the overall mortality rate for Fiscal Year 2020 (FY20) was 6.0%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance costs.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A census decrease results in an increase in unused space at all of the NIH-supported facilities and, thus while the need for maintenance still arises there is no need for new construction. There were no construction or renovation costs during FY20.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International and thus provide care that goes above and beyond that required by law. NIH is committed to maintaining the high quality of care and welfare at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility will vary over the course of a year and from year to year. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates, where expensive expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the costs per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a facility. This can be partially compensated by decreasing staff by a certain percentage as the chimpanzee population decreases (this will vary between facilities). Currently, animals are being relocated to the Federal Sanctuary at Chimp Haven, so NIH expects to see the cost efficiency begin to optimize at Chimp Haven while the efficiency at other facilities will decrease. Relocation efforts will continue into future years.
Based on the census as of October 1, 2020 and the most recent FY20 awards and payments, NIH spent a total of $6.16M for 436 animals for an average of $38.71 per day per chimpanzee across all facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR); 2) the Southwest National Primate Research Center (SNPRC); 3) the Alamogordo Primate Facility (APF); and 4) the Federal Sanctuary facility operated by Chimp Haven, Inc.
TABLE: FY20 Funds Provided for NIH-Owned and NIH-Supported Chimpanzees* as of October 1, 2020
|
Facilities |
||||
|---|---|---|---|---|
| Facility | Total # of chimpanzees | Total NIH Cost**, $M/year | Average NIH Cost***, $/animal/day | # of chimpanzees not to be relocated**** |
| KCCMR | 54 | 0.00 | $0.00× | Under Review |
| SNPRC | 53 | 0.60 | $31.02×× | Under Review |
| APF | 37 | 1.76 | $130.32××× | 37 |
| Chimp Haven | 292 | 3.80 | $35.65×××× | Not Applicable |
| Total | 436 | 6.16 |
Overall $38.71 |
Not Applicable |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned and NIH funds their cost of care
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
****Determined by the NIH Veterinary Panel (Information on the NIH Veterinary Panel Activities can be viewed at https://dpcpsi.nih.gov/sites/default/files/Veterinary_Panel_Meeting_Report_9-12-2019_508.pdf).
×No NIH funding was provided in FY20 for support for these animals as explained under “Current Costs Per Facility”.
××The NIH funding for FY20 provides only partial support for these animals as explained under “Current Costs Per Facility”.
×××The NIH funding for FY20 provides partial support for the animals as explained under “Current Costs Per Facility” (calculated for the periods 09/26/2019 – 07/30/2020 and 07/31/2020 – 01/31/2021).
××××The NIH funding for FY20 provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH Owned or Supported Chimpanzees
October 1, 2020 report
- Total number of animals owned or supported = 436
- Total cost for FY20 = $6.16M
- Total cost per animal per day = $38.71
October 18, 2019 report
- Total number of animals owned or supported = 463
- Total cost for FY19 = $7.59M
- Total cost per animal per day = $44.91
October 1, 2018 report
- Total number of animals owned or supported = 494
- Total cost for FY18 = $8.72M
- Total cost per animal per day = $48.36
October 1, 2017 report
- Total number of animals owned or supported = 518
- Total cost for FY17 = $9.62M
- Total cost per animal per day = $50.88
October 24, 2016 report
- Total number of animals owned or supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 21, 2015 report
- Total number of animals owned or supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 27, 2014 report:
- Total number animals owned or supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 27, 2013 report:
- Total number of animals owned or supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR (U42) - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- No funds were obligated in FY20 as the U42 award is in a no cost extension (07/01/2020 to 11/30/2020) and is carrying forward an unobligated balance that will support the care of 54 animals, with any additional costs incurred being covered by FY21 funds through a contract (to be awarded in November 2020).
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- The total cost to NIH for chimpanzee care from NOA dated 05/01/2020 - 04/30/2021 = $602,534.
- Under the P51 award, costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 53 chimpanzees owned by the SNPRC are partially supported with funds from NIH with the remaining costs provided by the Texas Biomedical Research Institute.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- The total cost to NIH for FY20 under the contract with Charles River Laboratories is $1,755,854.
- $650,000 was obligated in FY20 to cover costs incurred during the remaining 6 months for the base year (09/26/2019 – 07/30/2020).
- $1,105,854 was obligated in FY20 to initially cover projected costs for the first 6 months of option year one (07/31/2020 - 01/31/2021), with future costs for the remaining 6 months for this option year (02/01/2021 – 07/30/2021) and any additional costs incurred during the first 6 months of the next option year (07/31/2021 – 01/31/2022) to be covered by FY21 funds.
Chimp Haven, Inc. - A not-for-profit company that operates the federal sanctuary in Keithville, LA under contract with the NIH
- NIH-incurred costs under the contract with Chimp Haven, Inc. for period 09/30/2020- 06/30/2021 = $3,800,000, with future costs for the remaining 3 months for this option year (07/01/2021 – 09/29/2021) and any additional costs incurred during the first 6 months of the next option year (09/30/2021 -03/31/2022) to be covered by FY21 funds.
- NIH funding for the care of chimpanzees at the federal sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 18, 2019
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. For example, the overall mortality rate for Fiscal Year 2019 (FY19) was 6.3%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance costs.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A census decrease results in an increase in unused space at all of the NIH-supported facilities and, thus while the need for maintenance still arises there is no need for new construction. There were no construction or renovation costs during FY19.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International and thus provide care that goes above and beyond that required by law. NIH is committed to maintaining the high quality of care and welfare at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility will vary over the course of a year and from year to year. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates, where expensive expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the costs per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a facility. This can be partially compensated by decreasing staff by a certain percentage as the chimpanzee population decreases (this will vary between facilities). Currently, animals are being relocated to the Federal Sanctuary at Chimp Haven, so NIH expects to see the cost efficiency begin to optimize at Chimp Haven while the efficiency at other facilities will decrease. Relocation efforts will continue into future years.
Based on the census as of October 18, 2019 and the most recent FY19 awards and payments, NIH spent a total of $7.59M for 463 animals for an average of $44.91 per day per chimpanzee across all facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR); 2) the Southwest National Primate Research Center (SNPRC); 3) the Alamogordo Primate Facility (APF); and 4) the Federal Sanctuary facility operated by Chimp Haven, Inc.
TABLE: FY19 Funds Provided for NIH-Owned and NIH-Supported Chimpanzees* as of October 18, 2019
| Facility | Total # of chimpanzees |
Total NIH cost**, $M/year |
Average NIH Cost***,$/animal/day | # of Chimpanzees not to be Relocated**** |
|---|---|---|---|---|
| KCCMR | 63 | 1.76 | $76.54 | Under Review |
| SNPRC | 71 | 0.60 | $23.15x | Under Review |
| APF | 44 | 0.89 | $55.42xx | 44 |
| Chimp Haven | 285 | 4.34 | $41.72xxx | Not Applicable |
| Total | Total 463 |
Total 7.59 |
Overall $44.91 |
Not Applicable |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned and NIH funds their cost of care
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
****Determined by the NIH Veterinary Panel (Information on the NIH Veterinary Panel Activities can be viewed at https://dpcpsi.nih.gov/sites/default/files/Veterinary_Panel_Meeting_Rep…).
×The NIH funding provides only partial support for these animals as explained under “Current Costs Per Facility”.
××The NIH funding provides partial support for the animals as explained under “Current Costs Per Facility” (calculated for the period 09/26/2019 – 02/29/2020).
×××The NIH funding provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH Owned or Supported Chimpanzees
October 18, 2019 report
- Total number of animals owned or supported = 463
- Total cost for FY18 = $7.59M
- Total cost per animal per day = $44.91
October 1, 2018 report
- Total number of animals owned or supported = 494
- Total cost for FY18 = $8.72M
- Total cost per animal per day = $48.36
October 1, 2017 report
- Total number of animals owned or supported = 518
- Total cost for FY17 = $9.62M
- Total cost per animal per day = $50.88
October 24, 2016 report
- Total number of animals owned or supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 21, 2015 report
- Total number of animals owned or supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 27, 2014 report:
- Total number animals owned or supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 27, 2013 report:
- Total number of animals owned or supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR (U42) - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from Notice of Award (NOA) dated 09/01/2019 - 06/30/2020 = $1,756,360
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- The total cost to NIH for chimpanzee care from NOA dated 05/01/2019 - 04/30/2020 = $602,534.
- Under the P51 award, costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 71 chimpanzees owned by the SNPRC are partially supported with funds from NIH with the remaining costs provided by the Texas Biomedical Research Institute.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- The total cost to NIH for FY19 under the contract with Charles River Laboratories for period 09/26/2019 - 07/30/2020 = $886,230.
- This cost was obligated in FY19 to initially cover projected costs for the first 5 months of this option year (09/26/2019 - 02/29/2020), with future costs for the remaining 5 months for this option year (03/01/2020 – 07/30/2020) and any additional costs incurred during the first 5 months to be covered by FY20 funds.
Chimp Haven, Inc. - A not-for-profit company that operates the federal sanctuary in Keithville, LA under contract with the NIH
- NIH-incurred costs under the contract with Chimp Haven, Inc. for period 09/30/2019- 09/29/2020 = $3,913,880.
- NIH-incurred additional costs under the contract with Chimp Haven, Inc. for period 10/1/2018 to 9/30/2019 to extend contract support from 250 animals to up to 300 animals = $403,549
- NIH-incurred a one-time cost under the contract with Chimp Haven, Inc. for period 10/1/2018 to 9/30/2019 to purchase equipment related to veterinary care = $23,596
- Total cost under the contract for FY19 = $3,913,880 + $403,549 + $23,596 = $4,341,025
- NIH funding for the care of chimpanzees at the federal sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 1, 2018
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. For example, the overall mortality rate for Fiscal Year 2018 (FY18) was 4.6%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance costs.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A census decrease results in an increase in unused space at all of the NIH-supported facilities and, thus while the need for maintenance still arises there is no need for new construction. There were no construction or renovation costs during FY18.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International and thus provide care that goes above and beyond that required by law. NIH is committed to maintaining the high quality of care and welfare at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility will vary over the course of a year and from year to year. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates, where expensive expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the costs per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a facility. This can be partially compensated by decreasing staff by a certain percentage as the chimpanzee population decreases (this will vary between facilities). Currently, animals are being relocated to the Federal Sanctuary at Chimp Haven, so NIH expects to see the cost efficiency begin to optimize at Chimp Haven while the efficiency at other facilities will decrease. Relocation efforts will continue into future years.
Based on the census as of October 1, 2018 and the most recent FY18 awards and payments, NIH spent a total of $8.72 M for 494 animals for an average of $48.36 per day per chimpanzee across all facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR); 2) the Southwest National Primate Research Center (SNPRC); 3) the Alamogordo Primate Facility (APF) and; 4) the Federal Sanctuary facility operated by Chimp Haven, Inc.
FY18 Funds Provided for NIH Owned and Supported Chimpanzees*
Research Facilities
| Facility | # of Chimpanzees, as of 10/1/18 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| KCCMR | 127 | 2.61 | 56.30 |
| SNPRC | 77x | 0.60 | 21.35 |
| APF | 53 | 2.08 | 107.52 |
| Subtotal Eligible for Retirement | 257 | 5.29 | 56.55 |
| Chimp Haven | 237xx | 3.43 | 39.65 |
| Total 494 |
Total 8.72 |
Overall $48.36 |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned and NIH funds their cost of care
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
×The NIH funding provides only partial support for these animals.
××The NIH funding provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH Owned or Supported Chimpanzees
October 1, 2018 report
- Total number of animals owned or supported = 494
- Total cost for FY18 = $8.72M
- Total cost per animal per day = $48.36
October 1, 2017 report
- Total number of animals owned or supported = 518
- Total cost for FY17 = $9.62M
- Total cost per animal per day = $50.88
October 24, 2016 report
- Total number of animals owned or supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 21, 2015 report
- Total number of animals owned or supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 27, 2014 report:
- Total number animals owned or supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 27, 2013 report:
- Total number of animals owned or supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
Current Costs Per Facility
KCCMR (U42) - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from Notice of Award (NOA) dated 09/01/2018 - 08/31/2019 = $2,609,512
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- The total cost to NIH for chimpanzee care from NOA dated 05/01/2018 - 04/30/2019 = $602,534.
- Under the P51 award, costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 77 chimpanzees owned by the SNPRC are partially supported with funds from NIH with the remaining costs provided by the Texas Biomedical Research Institute.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- Total cost under the contract with Charles River Laboratories for period 09/26/2018 - 09/25/2019 = $2,076,558.
- Chimpanzees at APF began relocating to Chimp Haven in October 2016.
Chimp Haven, Inc. - A not-for-profit company that operates the federal sanctuary in Keithville, LA under contract with the NIH
- NIH-incurred costs under the contract with Chimp Haven, Inc. for period 09/30/2018- 09/30/2019 = $3,428,463.
- NIH funding for the care of chimpanzees at the federal sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 1, 2017
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. For example, the overall mortality rate for Fiscal Year 2017 (FY17) was 4.4%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance costs. For any facility, the cost of care per animal per day generally will decrease as the number of animals increase and vice versa.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A census decrease results in an increase in unused space at all of the NIH-supported facilities and, thus while the need for maintenance still arises there is no need for new construction. There were no construction or renovation costs during FY17.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International and thus provide care that goes above and beyond that required by law. NIH is committed to maintaining the high quality of care and welfare at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility will vary over the course of a year and from year to year. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates, where expensive expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the costs per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a facility. This can be partially compensated by decreasing staff by a certain percentage as the chimpanzee population decreases (this will vary between facilities). Currently, animals are being relocated to the Federal Sanctuary at Chimp Haven, so NIH expects to see the cost efficiency begin to optimize at Chimp Haven while the efficiency at other facilities will decrease. Relocation efforts will continue into future years.
Based on the census as of October 1, 2017 and the most recent FY17 awards and payments, NIH spent a total of $9.62 M for 518 animals for an average of $50.88 per day per chimpanzee across all facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR); 2) the Southwest National Primate Research Center (SNPRC); 3) the Alamogordo Primate Facility (APF) and; 4) the Federal Sanctuary facility operated by Chimp Haven, Inc. For FY16 the comparable numbers are a total of $9.06M for 542 animals for an average of $45.80 per day per chimpanzee.
FY17 Funds Provided for NIH Owned and Supported Chimpanzees*
Research Facilities
| Facility | # of Chimpanzees, as of 10/1/17 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| KCCMR | 131 | 2.48 | 51.87 |
| SNPRC | 80x | 0.572 | 19.59 |
| APF | 99 | 2.87 | 79.42 |
| Subtotal Eligible for Retirement | 310 | 5.92 | 52.32 |
| Chimp Haven | 208xx | 3.70 | 48.74 |
| Total 518 |
Total 9.62 |
Overall $50.88 |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: Chimpanzees are privately owned and NIH funds their cost of care
**Information in the table is based on data collected at the end of the fiscal year.
***The number of animals and thus the costs per animal per day will vary throughout the year at all facilities.
×The NIH funding provides only partial support for these animals.
××The NIH funding provides only partial support for these animals as per the CHIMP Act.
Cost Determinations
Total Costs Per Year for the NIH Owned or Supported Chimpanzees
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.50
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.60
October 27, 2013 report:
- Total number of animals owned or supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.50
October 27, 2014 report:
- Total number animals owned or supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 21, 2015 report
- Total number of animals owned or supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 24, 2016 report
- Total number of animals owned or supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.80
October 1, 2017 report
- Total number of animals owned or supported = 518
- Total cost for FY17 = $ 9.62M
- Total cost per animal per day = $50.88
Current Costs Per Facility
KCCMR (U42) - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from Notice of Award (NOA) dated 09/01/2017 - 08/31/2018= $2,479,036
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- The total cost to NIH for chimpanzee care from NOA dated 05/01/2017 - 04/30/2018 = $572,407.
- Under the P51 award, costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 80 chimpanzees owned by the SNPRC are partially supported with funds from NIH with the remaining costs provided by the Texas Biomedical Research Institute.
APF - Alamogordo Primate Facility, Alamogordo, NM is operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- Total cost under the contract with Charles River Laboratories for period 09/26/2017 - 09/25/2018 = $2,873,410.
- Chimpanzees at APF began relocating to Chimp Haven in October 2016.
Chimp Haven, Inc. - A not-for-profit company that operates the federal sanctuary in Keithville, LA under contract with the NIH
- NIH-incurred costs under the contract with Chimp Haven, Inc. for period 07/16/2017 - 09/30/2018 = $3,697,841.
- NIH funding for the care of chimpanzees at the federal sanctuary is made through a contract with matching requirements—contractor (Chimp Haven, Inc.) must match 1 dollar for every 3 dollars for operating expenses.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 24, 2016
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. For example, the overall mortality rate for Fiscal Year 2016 (FY16) was 5.4%, which is within the expected range. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance costs. For any facility, the cost of care per animal per day will decrease as the number of animals increases and vice versa.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. A census decrease results in an increase in unused space at all of the NIH-supported facilities and, thus while the need for maintenance still arises there is no near-term need for new construction. There were no significant construction or renovation costs during FY16.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) comply with local, state and federal laws that regulate animal research and also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International and thus provide care that goes above and beyond that required by law. NIH is committed to maintaining the high quality of care and welfare at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility can vary from year to year. These cost per day calculations are useful for monitoring efficiency within a facility but have limited usefulness when comparing across facilities. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates where expensive expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the cost per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a facility. This can be partially compensated by decreasing staff as the chimpanzee population decreases by a certain percentage (this will vary between facilities). Currently, all animals are beginning to relocate to the Federal Sanctuary at Chimp Haven so NIH expects to see the cost efficiency begin to optimize at Chimp Haven while the efficiency at other facilities will decrease.
Based on the census as of October 24, 2016 and the most recent FY16 awards and payments, NIH spent a total of $9.06 M for 542 animals for an average of $45.8 per day per chimpanzee across all facilities including: 1) the Keeling Center for Comparative Medicine and Research (KCCMR); 2) the Southwest National Primate Research Center (SNPRC); 3) the Alamogordo Primate Facility (APF) and; 4) the Federal Sanctuary facility operated by Chimp Haven, Inc. For FY15 the comparable numbers are a total of $10.19M for 573 animals for an average of $48.77 per day per chimpanzee.
FY16 Funds Provided for NIH Owned and Supported Chimpanzees*
Research Facilities
| Facility | # of Chimpanzees, as of 10/24/16 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| KCCMR | 135 | 2.61 | 53.00 |
| SNPRC, P51 | 81 | 0.602 | 20.40 |
| SNPRC, U42 | 0 | NA | NA |
| APF | 138 | 3.00 | 59.56 |
| Subtotal Eligible for Retirement | 354 | 6.21 | 48.08 |
| Chimp Haven | 188 | 2.85 | 41.55 |
| Total 542 |
Total 9.06 |
Overall 45.8 |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: chimpanzees are privately owned and NIH funds their cost of care
**Information in the tables is based on data collected at the end of the fiscal year.
***The number of animals and thus the cost per animal per day will vary throughout the year at all facilities.
Cost Determinations
Total Costs per Year for the NIH-Owned or -Supported Chimpanzees
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.5
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.6
October 27, 2013 report:
- Total number of animals owned or supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.5
October 27, 2014 report:
- Total number animals owned or supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 21, 2015 report
- Total number of animals owned or supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
October 24, 2016 report
- Total number of animals owned or supported = 542
- Total cost for FY16= $9.06M
- Total cost per animal per day = $45.8
Current Costs per Facility
KCCMR (U42) - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from Notice of Award (NOA) dated 09/01/2016 - 08/31/2017= $2,609,512
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- The total cost to NIH for chimpanzee care from NOA dated 05/18/2016 - 04/30/2017 = $602,534.
- Under the P51, costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 81 chimpanzees are owned by the SNPRC are partially supported with funds from NIH and the remainder provided by the Texas Biomedical Research Institute.
SNPRC (U42) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- The last of the NIH owned chimpanzees supported under the U42 at SNPRC were relocated to Chimp Haven in May 2016.
- No additional funds were provided to the SNPRC U42.
APF - Alamogordo Primate Facility, Alamogordo, NM and operated by Charles River Laboratories (a for-profit company) under contract with the NIH.
- Total cost for the base period under the contract with Charles River Laboratories for period 09/26/2016 - 09/25/2017 = $2,997,563.
- Chimpanzees at APF began relocating to Chimp Haven in October 2016.
Chimp Haven, Inc. - A not-for-profit company that operates the federal sanctuary in Keithville, LA under contract with the NIH.
- Total cost under the contract with Chimp Haven, Inc. for period 07/16/2016 - 07/15/2017 = $2,850,977.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 21, 2015
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. The number of animals at different facilities may also increase or decrease as a result of relocation. There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance costs. For any facility, the cost of care per animal per day will decrease as the number of animals increases and vice versa.
Facility Construction Costs
The federally supported chimpanzee population has been decreasing each year since the late 1990s due to a continuing breeding moratorium and natural mortality. The overall mortality rate for 2014-2015 was 5.6%, which is within the expected range. This census decrease has resulted in an increase in unused space at all of the NIH-supported facilities and, thus while the need for maintenance occasionally arises there is no near-term need for new construction; there are approximately 160 spaces available at the current NIH supported facilities. There were no significant construction or renovation costs during FY 2015.
Chimpanzee Care and Welfare
All federally supported chimpanzee facilities (sanctuary and non-sanctuary) not only comply with local, state and federal laws that regulate animal research but also are certified by the Association for Assessment and Accreditation of Laboratory Animal Care International and thus provide care that goes above and beyond that required by law. NIH is committed to maintaining the high quality of care and welfare at these facilities.
Since the number of chimpanzees at any facility will change over time, the cost per day per animal at a facility can vary from year to year. Also, if grant funds at a facility are not totally expended from the previous year, those funds are used to offset the current year’s cost and the cost per day per animal will appear lower; see TexBiomed U42 funding in the Tables below. These cost per day calculations are useful for monitoring efficiency within a facility but have limited usefulness when comparing across facilities. Costs vary between facilities due to the local economy, size of the chimpanzee population, and the presence of other nonhuman primates where expensive expertise may be shared. Generally, the larger the population at any facility (chimpanzees and other nonhuman primates), the lower the cost per day per animal at that facility; this reflects an economy of scale. In a non-breeding population, the total number of animals will eventually decrease to the point that an economy of scale can no longer be realized at a facility. This can be partially compensated by decreasing staff as the chimpanzee population decreases by a certain percentage (this will vary between facilities). However, over the next several years, even though the total cost of the chimpanzee program will continue to decrease, the efficiency will also decrease with a concomitant and unavoidable increase in the cost per day per animal.
Based on the census as of October 21, 2015 and the most recent FY15 awards and payments, NIH spent a total of $10.19M for 573 animals for an average of $48.77 per day per chimpanzee across all facilities including: 1) the two research facilities, Keeling Center for Comparative Medicine and Research (KCCMR) and the Southwest National Primate Research Center (SNPRC); 2) the Alamogordo Primate Facility (APF) and; 3) the federal sanctuary facility operated by Chimp Haven, Inc. For FY14 the comparable numbers are a total of $12.1M for 606 animals for an average of $54.66 per day per chimpanzee.
FY15 Funds Provided for NIH Owned and Supported Chimpanzees*
Research Facilities
| Facility | # of Chimpanzees, as of 10/21/15 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| KCCMR | 139 | 2.87 | 56.57 |
| SNPRC, P51 | 82 | 1.4 | 46.78 |
| SNPRC, U42 | 20 | 0 | 0 |
| Total | 241 | 4.27 | 48.54 |
Research Reserve Facility
| Facility | # of Chimpanzees, as of 10/21/15 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| APF | 149 | 3.16 | 58.10 |
Federal Sanctuary
| Facility | # of Chimpanzees, as of 10/21/15 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| Chimp Haven | 183 | 2.77M | 41.47 |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care.
- NIH-supported: chimpanzees are privately owned and NIH funds their cost of care.
**Information in the tables is based on data collected at the end of the fiscal year.
***The number of animals and thus the cost per animal per day will very throughout the year at all facilities.
Cost Determinations
Total Costs per Year for the NIH-Owned or -Supported Chimpanzees
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost for FY11 - $11.58M
- Total cost per animal per day = $44.5
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost for FY12 - $12.38M
- Total cost per animal per day = $50.6
October 27, 2013 preliminary report:
- Total number of animals owned or supported = 644
- Total cost for FY13 = $9.76M
- Total cost per animal per day = $41.5
October 27, 2014 report:
- Total number animals owned or supported = 606
- Total cost for FY14 = $12.09M
- Total cost per animal per day = $54.66
October 21, 2015 report
- Total number of animals owned or supported = 573
- Total cost for FY15= $10.19M
- Total cost per animal per day = $48.77
Current Costs per Facility
KCCMR (U42) - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from NOGA dated 09/01/2015 - 08/31/2016= $2,558,344
- 14 animals previously covered by a Division of Veterinary Resources contract were moved to the KCCMR U42 in FY14. Funds were provided to the U42 on 02/04/2015 to cover the care of these animals = $204,623 in NOGA dated 02/04-2012 – 08/31/2015. 13 animals were transferred to the federal sanctuary between December 2014 and April 2015.
- Funds to care for the DVR animals remaining at KCCMR were provided as indicated in the NOGA dated 09/04/2015 – 08/31/2016 = $104,313.
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- This is last year's total cost from NOGA dated 05/01/2015 - 04/30/2016, pro-rated for costs associated with chimpanzees only = $1.4M.
- Under the P51, costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 82 chimpanzees are owned by SNPRC and supported with funds from NIH.
SNPRC (U42) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Total cost from NOGA dated 08/01/2015 - 07/31/2016 = $1.00.
- 20 chimpanzees owned and supported by NIH.
- Unobligated FY14 funds were used to pay the FY 15 costs.
APF - Alamogordo Primate Facility, Alamogordo, NM and operated by Charles River Laboratories (a for-profit company) under contract with the NIH
- Total cost for the base period on contract HHSN276201400013C for period 09/26/2015 - 09/25/2016 = $3,155,644.
- Unlike the other facilities, the APF is located on a United States Air Force (USAF) base and thus operates under conditions of the contract between NIH and Charles River Laboratories which includes additional USAF-specific conditions detailed in a permit issued by the USAF.
- Use Permit No. USAF-ACC-KWRD-4-01-005 states that any chimpanzee designated as surplus must be removed from the APF premises.
- The same permit states that any chimpanzee removed from APF may not be returned to the APF. Further, no new animals may be introduced into the APF.
Chimp Haven, Inc.– A not-for-profit company that operates the federal sanctuary in Keithville, LA under contract with the NIH
- Total cost for the contract HHSN268201100065C for period 07/16/2015 - 07/15/2016 = $2,770,269.
- The Standards of Care for Chimpanzees Held in the Federally Supported Sanctuary System (42 CFR Part 9) and the CHIMP Act govern the activities at Chimp Haven.
- Space is filled as it becomes available. Chimp Haven received 13 animals from KCCMR between December 2014 and April 2015.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 27, 2014
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The total number of animals is expected to decrease annually as a result of normal mortality. The number of animals at different facilities may also increase or decrease as a result of relocation.
There are two categories of costs associated with the long-term care of chimpanzees owned or supported by NIH: (1) facility construction and (2) chimpanzee care and facility maintenance costs. For any facility, the cost of care per animal per day will increase as the number of animals decreases and vice versa.
Facility Construction Costs
The NIH supported chimpanzee facilities are specifically designed to accommodate the large size, strength, and the special behavioral requirements of this population. These living accommodations include indoor housing with air conditioning/heating, special wall furniture, and unrestricted access to large outdoor areas. The federally-supported chimpanzee population has been decreasing each year since the late 1990s due to a breeding moratorium and natural mortality. This census decrease has resulted in an increase in unused space at all of the NIH-supported facilities and, thus while the need for maintenance occasionally arises there is no near-term need for new construction; there are approximately 150 spaces available at the current NIH supported facilities. There were no significant construction or renovation costs during FY 2014.
Chimpanzee Care and Welfare
Based on the census as of October 27, 2014, and the most recent FY14 awards and payments, NIH spent a total of $12.1M for 606 animals for an average of $54.66 per day per chimpanzee across all facilities including: 1) the two research facilities, Keeling Center for Comparative Medicine and Research (KCCMR) and the Southwest National Primate Research Center (SNPRC); 2) the Alamogordo Primate Facility (APF);and 3) the federal sanctuary facility operated by Chimp Haven, Inc. For FY13 the comparable numbers are a total of $9.76M for 644 animals for an average of $41.50 per day per chimpanzee. There appears to be a disproportionately higher cost for fewer animals in FY14. This anomaly resulted from Chimp Haven receiving only a partial award ($744K) in FY 2013 because the cap on chimpanzee funding was not raised until FY 2014 by passage of the CHIMP Act Amendments of 2013. The remainder of the funds that would have been awarded to Chimp Haven in FY 2013 was paid with FY 2014 funds (see Tables and Cost per facility information below).
Since the number of chimpanzees at any facility will change over time, the “cost/day/animal” at that facility will vary over the course of a year. Also, as funds are provided to a facility in anticipation of receiving additional chimpanzees, the cost per day per animal may appear higher for that year; e.g., see the Chimp Haven funding in the Tables below. Conversely, if funds are not totally expended from the previous year, those funds are used to offset the current year’s cost and the cost per day per animal may appear lower; see TexBiomed funding in the Tables.
FY14 Funds Provided for NIH Owned and Supported Chimpanzees*
Research Facilities
| Facility | # of Chimpanzees, as of 10/27/14 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| KCCMR | 156 | 2.56 | 44.96 |
| SNPRC, P51 | 85 | 1.0 | 32.23 |
| SNPRC, U42 | 20 | 0 | 0 |
| Total | 261 | 3.56 | 37.37 |
Research Reserve Facility
| Facility | # of Chimpanzees, as of 10/27/14 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| APF | 154 | 4.09 | 60.36 |
Federal Sanctuary
| Facility | # of Chimpanzees, as of 10/27/14 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| Chimp Haven | 191 | 4.44M | 63.69 |
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: chimpanzees are privately owned and NIH funds their cost of care
**Information in the tables is based on data collected at the end of the fiscal year.
***The number of animals and thus the cost per animal per day will very throughout the year at all facilities.
Cost Determinations
Total per Year
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost per year - $11.58M
- Total cost per animal per day = $44.5
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost per year - $12.38M
- Total cost per animal per day = $50.6
October, 2013 preliminary report:
- Total number of animals owned or supported = 644
- Total cost per year FY13 = $9.76M
- Total cost per animal per day = $41.5
October 27, 2014 report:
- Total number animals owned or supported = 606
- Total cost per year = $12.09M
- Total cost per animal per day = $54.66
Cost per Facility
KCCMR (U42) - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from Notice of Grant Award (NOGA) dated 09/01/2014 - 08/31/2015= $2,560,584
- 14 animals previously covered by a NIH Division of Veterinary Resources contract are now covered under the grant above at no additional FY14 dollars.
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- This is last year's total cost from NOGA dated 05/01/2014 - 04/30/2015, pro-rated for costs associated with chimpanzees only = $1.0M.
- Under the P51, costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 85 chimpanzees are owned by SNPRC and supported with funds from NIH.
SNPRC (U42) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Total cost from NOGA dated 08/01/2014 - 07/31/2015 = $1.00.
- 20 chimpanzees owned and supported by NIH.
- Unobligated FY13 funds were used to pay the FY 14 costs.
APF - Alamogordo Primate Facility, Alamogordo, NM
- Cost to extend the previous contract from 06/30/2014 to 09/25/2014 = $934,984.
- Total cost for the base period on contract HHSN276201400013CC for period 09/26/2014 - 09/25/2015 = $3,150,452.
- Unlike the other facilities, the APF does not have an active chimpanzee research program; no invasive research is conducted on these chimpanzees while on the premises. According to contract solicitation NHLBI-CSB-(RR)-SS-2011-264-KJM (HHS, 2011c), the current agreements between the NIH and the U.S. Air Force (USAF) prescribe that no invasive research shall be conducted on chimpanzees currently held at the APF.
- Use Permit No. USAF-ACC-KWRD-4-01-005 states that any chimpanzee designated as surplus (eligible for retirement) must be immediately removed from the APF premises.
- The same permit states that any chimpanzee removed from APF for research may not be returned to the APF. Further, no new animals may be introduced into the APF.
Chimp Haven, Inc. - Operates the federal sanctuary in Keithville, LA
- Total cost for the contract HHSN268201100065C for period 07/16/2014 - 07/15/2015 = $2,734,593.
- In January 2014, $1.2M was provided to cover the FY13 short-funding.
- In April 2014, $504,396 was provided to cover the cost of the additional chimpanzees transferred from the New Iberia Research Center.
- The Standards of Care for Chimpanzees Held in the Federally Supported Sanctuary System (42 CFR Part 9) and the CHIMP Act govern the activities at Chimp Haven.
- Space is filled as it becomes available.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 27, 2013
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The number of animals is expected to decrease annually as a result of normal mortality.
There are two types of costs associated with the long-term care of chimpanzees owned or supported by NIH: appropriate facility construction and chimpanzee care and welfare costs.
Facility Construction Costs
Chimpanzee facilities are uniquely designed for these large animals, including indoor housing with air conditioning/heating, special wall furniture, and outdoor housing tailored to chimpanzee size and behavior. In 2002-2004 NIH spent approximately $11.8 million to construct a federal sanctuary system operated by Chimp Haven, Inc. to appropriately house 130 federally owned chimpanzees over their life time. Approximately $2 million was provided to the Texas Biomedical Research Institute in September 2010 to renovate facilities to increase the capacity of the Southwest National Primate Research Center.
Chimpanzee Care and Welfare
Based on the most recent awards and payments, NIH is spending an average of $44.50 per day per chimpanzee in research facilities; $61.3 per day per chimpanzee in the research reserve at Alamogordo Primate Facility (APF); and $42.70 per day per chimpanzee in the federal sanctuary facility operated by Chimp Haven, Inc. See Tables for detailed figures.
FY13 Funds Provided for NIH-Owned and -Supported Chimpanzees
Research Facilities (Preliminary)
| Facility | # of Chimpanzees, as of 10/27/13 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day (avg) |
|---|---|---|---|
| NIRC | 59 | 1.01 | 46.7 |
| KCCMR | 147 | 2.44 | 46.4 |
| KCCMR, DVR | 16 | .4 | 68.8 |
| SNPRC, P51 | 89 | .92 | 28.2 |
| SNPRC, U42 | 22 | .65 | 80.9 |
| Total | (333) | (5.42) | (44.5) |
Research Reserve Facility (Preliminary)
| Facility | # of Chimpanzees, as of 10/27/13 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| APF | 162 | 3.60 | 61.3 |
Federal Sanctuary
| Facility | # of Chimpanzees, as of 10/27/13 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| Chimp Haven | 149 | .7441 | 42.71 |
1Funds provided to last approximately 4 months at a spend rate of $190,000 per month. See explanation below in Cost Determinations.
*Definition of owned and supported:
- NIH-owned: NIH owns the chimpanzee and funds their cost of care
- NIH-supported: chimpanzees are privately owned and NIH funds their cost of care
**Cost Determinations - see below.
***Information in the tables is based on data collected at the end of the fiscal year. The number of animals and thus the cost per animal per day will very throughout the year at all facilities.
Cost Determinations
April 15, 2011 report:
- Total number animals owned or supported = 734
- Total cost per year = $11.81M
- Total cost per animal per day = $44.1
October 31, 2011 report:
- Total number of animals owned or supported = 713
- Total cost per year - $11.58M
- Total cost per animal per day = $44.5
October 23, 2012 report:
- Total number of animals owned or supported = 669
- Total cost per year - $12.38M
- Total cost per animal per day = $50.6
October, 2013 preliminary report:
- Total number of animals owned or supported = 644
- Total cost per year
- Total cost per animal per day
NIRC - New Iberia Research Center, Lafayette, LA
- Total cost from NOGA dated 09/16/2013 - 09/15/2014= $1,006,169
- All NIH-owned NIRC chimpanzees are permanently ineligible for biomedical research.
KCCMR - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from NOGA dated 09/01/2013 - 08/31/2014= $2,437,039
- Total cost for Division of Veterinary Resources (DVR) base contract HHSN263201200075I for period 8/7/2013 – 8/6/2014 = $401,967.20.
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- This is last year's total cost from NOGA dated 05/01/2012 - 04/30/2013, pro-rated for costs associated with chimpanzees only = $917,305.
- Most recent numbers are pending from the P51 where costs are shared between care for the chimpanzees and several thousand other non-human primates.
- 89 chimpanzees are owned by SNPRC and supported with funds from NIH.
SNPRC also owns 26 animals not supported by NIH
SNPRC (U42) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Total cost from NOGA dated 08/01/2013 - 07/31/2014 = $650,000. 22 chimpanzees owned and supported by NIH.
- The Tex Biomed cooperative agreement is funded to accommodate the potential influx of the research colony animals described by the Working Group. Until this colony location is determined the actual costs of animals on the cooperative agreement will not be accurate.
APF - Alamogordo Primate Facility, Alamogordo, NM
- Total cost for first option on contract HHSN268201200068C for period 06/30/2013 - 06/29/2014 = $3,624,969.
Chimp Haven, Inc. – Operates the federal sanctuary in Keithville, LA
- Total cost for base contract HHSN268201100065C for period 07/16/2013 - 07/15/2014 = $744,000.
- Funds provided are expected to last approximately 4 months at a spend rate of $190,000 per month.
Archive - Costs for Maintaining Humane Care and Welfare of Chimpanzees, Oct. 23, 2012
NIH conducts an annual census of its owned and supported chimpanzees after the end of each fiscal year (Sept. 30) to provide the public with official figures on their numbers and cost of care. The number of animals is expected to decrease annually as a result of normal mortality.
There are two types of costs associated with the long-term care of chimpanzees owned or supported by NIH: appropriate facility construction and chimpanzee care and welfare costs.
Facility Construction Costs
Chimpanzee facilities are uniquely designed for these large animals, including indoor housing with air conditioning/heating, special wall furniture, and outdoor housing tailored to chimpanzee size and behavior. In 2002-2004 NIH spent approximately $11.8 million to construct a federal sanctuary system operated by Chimp Haven, Inc. to appropriately house 130 federally owned chimpanzees over their life time. Approximately $2 million was provided to the Texas Biomedical Research Institute in September 2010 to renovate facilities to increase the capacity of the Southwest National Primate Research Center.
Chimpanzee Care and Welfare
Based on the most recent awards and payments, NIH is spending an average of $44 per day per chimpanzee in research facilities; $66 per day per chimpanzee in the research reserve facility at Alamogordo Primate Facility (APF); and $53 per day per chimpanzee in the federal sanctuary facility operated by Chimp Haven, Inc. The average for research facilities becomes $50 per day if the research reserve facility at APF is included. See Tables for detailed figures.
Changes in the Chimpanzee Management Plan since the Last Posting
The last posting, October 31, 2011, reported only those animals owned or supported by the former National Center for Research Resources (NCRR)*. The current posting reports on all animals owned or supported by NIH; this resulted in an increase in reporting of 16 animals owned and supported by the Division of Veterinary Resources (DVR) within the Office of Research Services at NIH. The DVR chimpanzees are housed at the Keeling Center for Comparative Medicine and Research (see Table below).
The October 31, 2011 posting reported 125 animals owned by the Southwest National Primate Research Center (SNPRC, Texas Biomedical Research Institute) and supported by NCRR. The current posting reports 91 animals owned by SNPRC and supported by NIH; SNPRC assumed support for the remaining SNPRC-owned animals (see details in Table below). National Primate Research Centers (NPRC) that own chimpanzees can choose to support their privately owned animals with non-federal funds.
On September 21, 2012, the NIH announced that it will be relocating the NIH-owned chimpanzees currently located at the New Iberia Research Center (NIRC) in New Iberia, La. All of the chimpanzees have been declared permanently ineligible for biomedical research and therefore, eligible for retirement. NIH will be retiring as many chimpanzees as can be accommodated by existing capacity within the Federal Sanctuary System and relocate the remainder to the SNPRC, Texas Biomedical Research Institute (Texas Biomed) until space within the Federal Sanctuary System becomes available. Through the existing Texas Biomed cooperative agreement, the NIH provided sufficient funds to support the relocation of chimpanzees to Texas Biomed and to support their care and maintenance once relocated (U42, see Table below). Any excess funds stemming from this award will be used to offset the costs to the NIH of any additional years remaining in the U42.
As of December 2011, no new research using chimpanzees has been funded.
* NCRR ceased to exist after the creation of the National Center for Advancing Translational Sciences (NCATS) on Dec. 23, 2011. The responsibility for chimpanzees formerly in NCRR’s care now lies with the NIH Division of Program Coordination, Planning, and Strategic Initiatives within the NIH Office of the Director.
Change in Overall Cost to the Government from Previous Reporting Period
October 31, 2011 report:
- Total number animals owned or supported = 713
- Total cost per year = $11.58M
- Total cost per animal per day = $44.5
October 23, 2012 report:
- Total number of animals owned or supported = 670
- Total cost per year - $12.38M
- Total cost per animal per day = $50.6
Cost of NIH-Owned and -Supported* Chimpanzees
Research Facilities
| Facility | # of Chimpanzees, as of 10/23/12 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| NIRC | 110 | 1.86 | 46.3 |
| KCCMR | 151 | 2.56 | 46.5 |
| KCCMR, DVR | 16 | .39 | 67.4 |
| SNPRC, P51 | 91 | .92 | 27.7 |
| SNPRC, U42 | 24 | .52 | 59.4 |
| Total | (392) | (6.24) | (43.6) |
Research Reserve Facility
| Facility | as of 10/23/12 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| APF | 169 | 4.04 | 65.5 |
Federal Sanctuary
| Facility | # of Chimpanzees, as of 10/23/12 (total) |
NIH Cost**, $M/year (total) |
NIH cost***, $/animal/day, (avg) |
|---|---|---|---|
| Chimp Haven | 109 | 2.10 | 52.8 |
*Definition of owned and supported:
NIH-owned: NIH owns the chimpanzee and funds their cost of care NIH-supported: chimpanzees are privately owned and NIH funds their cost of care
**Cost determined as below based on most recent 2012 to 2013 Notice of Grant Awards (NOGA) or invoices/obligated funds for contracts with Chimp Haven and APF.
***Information in the tables is based on data collected at the end of the fiscal year. The number of animals and thus the cost per animal per day will very throughout the year at all facilities.
NIRC - New Iberia Research Center, Lafayette, LA
- Total cost from NOGA dated 09/16/2012 - 09/15/2013= $1,858,987
- All NIH-owned NIRC chimpanzees are permanently ineligible for biomedical research.
KCCMR - Keeling Center for Comparative Medicine and Research, Bastrop, TX
- Total cost from NOGA dated 09/01/2012 - 08/31/2013= $2,564,160
- Total cost for Division of Veterinary Resources (DVR) base contract HHSN263201200075I for period 8/7/2012 – 8/6/2013 = $393,733.
SNPRC (P51) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Total cost from NOGA dated 05/01/2012 - 04/30/2013, pro-rated for costs associated with chimpanzees only = $917,305.
- 91 chimpanzees are owned by SNPRC and supported with funds from NIH.
SNPRC also owns 26 animals not supported by NIH.
SNPRC (U42) - Southwest National Primate Research Center (Texas Biomedical Research Institute), San Antonio, TX
- Total cost from NOGA dated 09/21/2012 - 07/31/2013= $3,347,851. This includes $523,117 for the current year support and a supplement of $142,850 for partial support of animals in the previous year.
- 24 chimpanzees owned and supported by NIH.
NOGA also includes $2,681,884 to cover the costs of moving 110 chimpanzees from NIRC and the care and maintenance of the animals as they arrive at the center. These animals are not yet located at SNPRC. This cost does not apply to the 24 animals already at the center.
APF - Alamogordo Primate Facility, Alamogordo, NM
- Total cost for first option on contract HHSN268201200068C for period 07/16/2012 - 07/15/2013 = $4,037,874.
Chimp Haven, Inc. – Operates the federal sanctuary in Keithville, LA
- Total cost for base contract HHSN268201100065C for period 06/30/2012 - 06/29/2013 = $2,097,120.
Archive - Chimpanzee Management Program 2013
The NIH Chimpanzee Management Program (ChiMP) supports long-term, cost-effective housing and maintenance at NIH-supported facilities for chimpanzees. ORIP provides programmatic oversight of the facilities and ensures they comply with the Animal Welfare Act, and policies concerning laboratory animal care and use.
In 1995, a breeding moratorium on NCRR-owned and supported chimpanzees was instituted. Soon after, the Chimpanzee Management Plan Working Group was created to periodically assess the need for chimpanzees in research and to report its findings to the National Advisory Research Resources Council, NCRR's advisory body. The Working Group serves as a fact-finding body that gathers information, analyzes relevant issues and facts, and drafts proposed position papers. The Working Group is comprised of non-government members with a wide range of scientific and non-scientific expertise.
The most recent meeting of the Working Group was held in March 2007 and the Report of the Chimpanzee Management Plan Working Group, (66K PDF, requires free Acrobat Reader Exit Disclaimer), was provided to the National Advisory Research Resources Council on May 22, 2007.
While NCRR acknowledges the continuing importance of chimpanzees to biomedical research, it also recognizes the fiduciary responsibilities it has to maintain the health and well-being of the chimpanzees already in its care. Since chimpanzees in captivity can live 50 years or more, high-quality care for just one animal over its lifespan can cost $300,000 to $500,000. In addition to its responsibilities to provide care for the existing chimpanzees owned or supported by NCRR, the Center must also fulfill its budget responsibilities to its other programs and resources, which are critical to the creation and delivery of lifesaving cures and treatments to the American people.
Therefore, after careful review of existing chimpanzee resources, NCRR has determined that it does not have the financial resources to support the breeding of chimpanzees that are owned or supported by NCRR. However, NCRR will continue to honor its commitments to the existing chimpanzee facilities, including the federal sanctuary for chimpanzees that are no longer needed in biomedical research.
Chimpanzee Sanctuary
In September 2002, NCRR announced the award of a contract to Chimp Haven, Inc Exit Disclaimer., a private, nonprofit organization, to establish and operate a chimpanzee sanctuary. The sanctuary will provide lifetime care for federally owned or supported chimpanzees that are no longer needed for biomedical research. Over the term of the 10-year, cost-sharing contract, NCRR will provide approximately $19 million in total costs, and Chimp Haven will contribute approximately $4 million toward direct costs. NCRR also awarded two construction grants, totaling a little over $11.5 million, so that Chimp Haven could develop and build the state-of-the-art facility. A sanctuary system to provide lifetime care of retired research chimpanzees was mandated by the Chimpanzees Health Improvement, Maintenance, and Protection (CHIMP) Act of December 2000.
In addition, NCRR has issued standards to implement provisions of the CHIMP Act, which authorized the Secretary of the Department of Health and Human Services to develop and publish standards of care for chimpanzees held in the sanctuary system. The standards apply to only those facilities receiving funds as a part of the federally-funded chimpanzee sanctuary system.
On July 8, 2008, the Chimpanzee Sanctuary Working Group toured the Chimp Haven facility with the Chimp Haven staff and Board of Directors. Following the tour, the Working Group and NIH staff met to review the on-site information provided by Chimp Haven. The Chimpanzee Sanctuary Working Group prepared a summary that was provided to the National Advisory Research Resources Council on September 16, 2008.
Contact Information
For further information, contact:
Sheri Hild, Ph.D.
Program Officer
Division of Comparative Medicine
Office of Research Infrastructure Programs
Division of Program Coordination, Planning, and Strategic Initiatives
Office of the Director
National Institutes of Health
One Democracy Plaza
6701 Democracy Boulevard, MSC 4874
Bethesda, Maryland 20892-4874 (20817 for express mail)
Phone: 301-435-0744
Fax: 301-480-3819
E-mail: Sheri Hild
Archive - Statement on Relocation of NIH Chimpanzees at New Iberia Research Center December 2012
Update (December 17, 2012): NIH issued the following announcement on its plans to move all of its chimpanzees from the New Iberia Research Center directly to the Federal Sanctuary System: https://stagetestdomain3.nih.gov/news-events/news-releases/nih-plans-relocate-its-chimpanzees-new-iberia-federal-sanctuary-system-0
December 3, 2012: Since the September 21, 2012, statement below, NIH has been collaborating extensively with Chimp Haven, NIRC, and other organizations that care about chimpanzees to identify the maximum number of chimpanzees that can go directly into the Federal Sanctuary System within the current funding and time constraints and taking into consideration the health of each individual chimpanzee. The process of relocating these chimpanzees will take time and care to ensure their continued well-being. NIH will continue to update the community as these numbers are finalized. NIH’s goal is to retire all 110 of its research-ineligible chimpanzees into the sanctuary as soon as possible.
September 21, 2012: The NIH today announced that it will be relocating the NIH-owned chimpanzees currently located at the New Iberia Research Center (NIRC) in New Iberia, La., because NIH funding to the facility will end in September 2013. This circumstance, combined with NIH’s anticipation that there will be a substantial reduction in the number of chimpanzees needed for research that meets the Institute of Medicine (IOM) criteria, prompted NIH Director Francis S. Collins, M.D., Ph.D., to designate all of these chimpanzees permanently ineligible for biomedical research. As of that date, there were 110 NIH-owned chimpanzees at NIRC.
Approximately 10 to 20 of the chimpanzees will be relocated to the federally supported chimpanzee sanctuary operated by Chimp Haven, Inc. in Keithsville, La., which would put Chimp Haven at or near full occupancy. NIH is continuing to evaluate options to move additional animals to Chimp Haven. It is expected that the remaining chimpanzees will be relocated to the Texas Biomedical Research Institute in San Antonio, Tx. Texas Biomedical has the specialized resources, experience, capacity, and funding mechanism to provide continued high-quality care for the chimpanzees. The animals being relocated to Texas Biomedical are considered permanently ineligible for biomedical research and therefore are retirement eligible. Relocation of the chimpanzees will be conducted on a timescale that will allow for optimal transition of each individual chimpanzee with careful consideration of their welfare, including their health and social grouping.
NIH will continue to keep the community informed throughout this transition which is expected to occur over several months.
Supercomputing Technologies Funded by ORIP Helped Spur Approval of COVID-19 Vaccines and Save Lives*
Supercomputing has been gaining attention for its applications across biomedical research. A type of high-performance computing...




