Reshaping One of Nature’s Most Complex Defense Mechanisms to Help Fight Diseases
Biomedical research with nonhuman primates (NHPs) has resulted in several notable medical breakthroughs.1–5 NHP research has been essential to our understanding of how the human body responds to infectious and noninfectious diseases and organ transplantation, and to finding therapies for certain cancers and AIDS. NHP biomedical research is made more translatable to human disease using NHP-related laboratory reagents for the characterization and modulation of disease-causing processes.
Established by a grant in 2000 from the Office of Research Infrastructure Programs at the National Institutes of Health, the Nonhuman Primate Reagent Resource (NHPRR) program functions as a one-stop-shop for NHP laboratory reagents. NHPRR’s mission is to facilitate the optimal use of NHP models in biomedical research by providing reagents to increase precision and predictability of the NHP model. The high-quality reagents provided through NHPRR (useful for NHP or human studies) are manufactured by MassBiologics, a nonprofit company at the University of Massachusetts Medical School. Most of the reagents available through NHPRR for research are classified as “biologics,” which are genetically engineered proteins derived from genetic sequences. In addition to research applications, biologics are being used in humans to treat infectious illnesses, autoimmune diseases, and some cancers.
The NHPRR has a collection of novel biologics or antibodies that deplete certain cell populations or perturb cell communication pathways important for the immune response. The cells targeted by biologics serve to mount an immune response to defend against invading bacteria, viruses, or toxins. However, in some cases, the reagents will help show that activation of the targeted cells is actually responsible for promoting disease. In essence, these reagents can identify and reshape elements of a complex defense mechanism to help fight complex diseases.
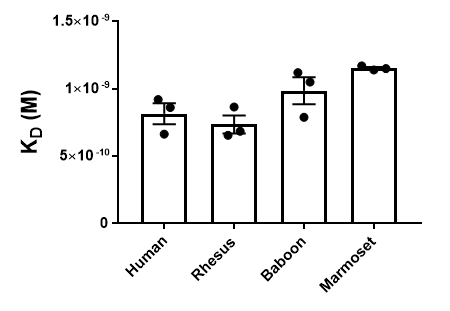
One type of reagent (a depletion antibody) targets the molecule CD40 to block the activation of certain T cell populations. In some human conditions, blocking the abnormal activation of these T cells may prevent a harmful immune response, such as organ transplant rejection.6,7 Other biologics that are available through NHPRR are called “bispecific” antibodies that simultaneously bind to two targets to enhance the destruction of certain cell populations (e.g., cancer cells).8 Bispecific antibodies can also be designed to carry therapeutics across the blood-brain-barrier (an almost insurmountable barrier) of the body and will facilitate the treatment and ultimately the cure for many brain-related illnesses.9
The ultimate goal of the NHPRR program is to develop reagents that are applicable for clinical use. Currently, NHPRR has several reagents that are being tested as new disease intervention drugs. For more information on the program and to view the current catalog of reagents, visit their website at www.nhpreagents.org.
References
1 Bontrop RE. Nonhuman primates: essential partners in biomedical research. Immunological Review. 2001;183:5–9.
2 Capitanio JP, Emborg ME. Contributions of nonhuman primates to neuroscience research. Lancet. 2008;371:1126–1135.
3 Haigwood N (2016). The critical role of nonhuman primates in medical research (white paper).
4 Enders J, Weller T, Robbins F. Cultivation of the Lansing strain of poliomyelitis virus in cultures of various human embryonic tissues. Science. 1949;109(2822):85–87.
5 Anderson DJ and Kirk AD. Primate models in organ transplantation. Cold Spring Harbor Perspectives in Medicine. 2013; 3(9):a015503.
6 Zhai Y and Kupiec-Weglinski JW. The CD154-CD40 costimulation pathway in organ transplantation. Transplantation Reviews. 2004;18(1):10–19.
7 Mohiuddin MM, Singh AK, Corcoran PC, Thomas III ML, Clark T, Lewis BG, et al. Chimeric 2C10R4 anti-CD40 antibody therapy is critical for long-term survival of GTKO.hCD46.hTBM pig-to-primate cardiac xenograft. Nature Communications. 2016; 5(7):11138.
8 Kantarjian H, Stein A, Gökbuget N, Fielding AK, Schuh AC, Ribera JM, et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. The New England Journal of Medicine. 2017; 76(9):836–847.
9 Merriam, D, Chen, C, Méndez-Lagares, G, Michaels, AJ, Yan, J, Casaz, P, Reimann, KA, et al. Depletion of gut-resident CCR5+ cells for HIV cure strategies. AIDS Research and Human Retroviruses. 2017;33(S1):S70–S80.



